| |
NS5A Resistance-Associated Substitutions in Patients with Genotype 1 Hepatitis C Virus: Prevalence and Effect on Treatment Outcome
|
| |
| |
Download the PDF here
Article In Press Jnl of Hepatology Jan 17 2017
Stefan Zeuzem, Masashi Mizokami, Stephen Pianko, Alessandra Mangia, Kwang-Hyub Han, Ross Martin, Evguenia Svarovskaia, Hadas Dvory-Sobol, Brian Doehle, Charlotte Hedskog, Chohee Yun, Diana M. Brainard, Steven Knox, John G. McHutchison, Michael D. Miller, Hongmei Mo, Wan-Long Chuang, Ira Jacobson, Gregory J. Dore, Mark Sulkowski
Abstract
Background
The efficacy of NS5A inhibitors for the treatment of patients chronically infected with hepatitis C virus (HCV) can be affected by the presence of NS5A resistance-associated substitutions (RASs). We analyzed data from 35 phase 1, 2, and 3 studies in 22 countries to determine the pretreatment prevalence of various NS5A RASs, and their effect on outcomes of treatment with ledipasvir-sofosbuvir in patients with genotype 1 HCV.
Methods
NS5A gene deep sequencing analysis was performed on samples from 5,397 patients in Gilead clinical trials. The effect of baseline RASs on sustained virologic response (SVR) rates was assessed in the 1,765 patients treated with regimens containing ledipasvir-sofosbuvir.
Results
Using a 15% cut-off, pretreatment NS5A and ledipasvir-specific RASs were detected in 13% and 8% of genotype 1a patients, respectively, and in 18% and 16% of patients with genotype 1b. Among genotype 1a treatment-naïve patients, SVR rates were 91% (42/46) vs 99% (539/546) with and without ledipasvir-specific RASs, respectively. Among treatment-experienced genotype 1a patients, SVR rates were 76% (22/29) vs 97% (409/420) with and without ledipasvir-specific RASs, respectively. Among treatment-naïve genotype 1b patients, SVR rates were 99% for both those with and without LDV-specific RASs (71/72 vs 331/334), and among treatment-experienced genotype 1b patients, SVR rates were 89% (41/46) vs 98% (267/272) for those with and without ledipasvir-specific RASs, respectively.
Conclusions
Pretreatment ledipasvir-specific RASs that were present in 8%-16% of patients have an impact on treatment outcome in some patient groups in particular treatment-experienced patients with genotype 1a HCV.
Lay summary
The efficacy of treatments using NS5A inhibitors for patients with chronic Hepatitis C virus (HCV) infection can be affected by the presence of NS5A resistance-associated substitutions (RASs). We reviewed results from 35 clinical trials where patients with genotype 1 HCV infection received treatments that included ledipasvir-sofosbuvir to determine how prevalent NS5A RASs are in patients at baseline, and found that ledipasvir-specific RASs were present in 8-16% of patients prior to treatment and had a negative impact on treatment outcome in subset of patient groups in particular treatment-experienced patients with genotype 1a HCV.
INTRODUCTION
Due to high rates of viral replication and an error prone HCV RNA polymerase, tremendous variability of HCV has been observed within infected patients (quasispecies) with all single mutations that do not abolish viral replication thought to be pre-existing [1]. As a result, NS5A RASs are observed at baseline in patients infected with chronic HCV. Deep sequencing enables detection of HCV substitutions, point deletions, or insertions within the quasispecies down to a frequency of 1%. However, commercially available assays based on standard population HCV sequencing or not cross-validated next generation, also called deep sequencing, report variants with a frequency of ⩾15% of the quasispecies.
The prevalence of baseline NS5A RASs has been reported to be 6% to 16% using population sequencing (cut off 15-25%) or deep sequencing (cut off 1%) [[2], [3], [4]].
Interestingly, the prevalence and type of baseline NS5A RASs may vary by geographic regions. For example, the prevalence of the NS5A M28V in genotype 1a-infected patients was shown to be higher in the United States than in Europe, 7% versus 0%, respectively [5]. Furthermore, the prevalence of genotype 3 NS5A Y93H varied between 0% and 17% in different geographic regions [6]. A comparison of baseline prevalence of RASs in Japanese and Western patients showed that the prevalence of Q80L and S122G in NS3, and L28M, R30Q and Y93H in NS5A was significantly higher in Japanese patients than the Western counterparts [7].
Many currently approved interferon (IFN)-free regimens for the treatment of chronic hepatitis C (HCV) include an inhibitor of HCV NS5A. To date, there are five NS5A inhibitors approved for treatment of chronic HCV infection; ledipasvir (LDV), daclatasvir, and velpatasvir (which are all administered with the NS5B inhibitor sofosbuvir), and ombitasvir (in a fixed-dose combination with the protease inhibitor paritaprevir, the nonnucleoside NS5B polymerase inhibitor dasabuvir, and ritonavir, a potent inhibitor of CYP3A4 enzymes), and elbasvir (in a fixed-dose combination with the protease inhibitor grazoprevir) [[8], [9], [10], [11], [12]]. The presence of baseline NS5A RASs may impact treatment outcome of some NS5A inhibitor containing HCV regimens due to the intrinsic qualities of the NS5A inhibitor, drug pharmacology, or effects of the other compounds within the treatment regimen. However, depending on how NS5A RASs are defined and included in resistance analysis, as well as what level of variant detection is utilized, different results may be obtained. To date, three definitions of NS5A variants that are associated with resistance have been used most commonly; polymorphisms at RAS positions (RAPs), class RASs, and drug-specific RASs. Polymorphisms at RAS positions are defined as any change from reference sequence for a specific genotype at positions associated with NS5A inhibitor resistance. NS5A class RASs are substitutions that have been shown to emerge on treatment or confer a significant reduction in susceptibility in vitro (e.g., >2.5 fold change in EC50) to any approved or investigational NS5A inhibitor. Drug-specific RASs refer to substitutions that have been shown to emerge on the specific drug treatment or confer significantly reduced susceptibility in vitro to the specific NS5A inhibitor. In addition, drug-specific RASs can be categorized into groups with different levels of reduced susceptibility to the drug.
To enable comparisons of resistance analyses between clinical trials, standardization of RAS definitions and sensitivity cut offs is needed. In several studies, population sequences were used for resistance analysis (cut off of variant detection 15%-25%) and NS5A polymorphisms at RAS positions were defined as RAPs. In these studies, the presence of baseline NS5A polymorphisms at RAS positions had shown no significant impact on treatment outcome [[5], [12]]. Further study is needed to understand the role of RASs present at frequencies below 15% and whether substitutions without an in vitro susceptibility change to the NS5A inhibitor may dilute a clinical signal by RASs that do confer reduced susceptibility to a specific NS5A inhibitor.
Here, we characterized the prevalence of baseline NS5A RASs in 5,397 NS5A inhibitor-naïve patients infected with genotype 1a or 1b HCV according to geographic regions. Moreover, we assessed the effect of baseline NS5A RASs, defined as NS5A RAPs, NS5A class RASs or LDV-specific RASs using 1% and 15% sensitivity of substitution detection cut offs, on treatment outcome among 1,765 patients treated with currently recommended regimens containing ledipasvir-sofosbuvir. A previous analysis using a portion of the same dataset has recently been published [13]. That analysis concerned the prevalence and effect on treatment of NS3, NS5A, and NS5B RASs, and included data on patients who had been treated with regimens/durations that have not been incorporated into label recommendations or treatment guidelines. The current study covers only NS5A RASs and includes data only from patients who received guideline-recommended regimens.
MATERIAL AND METHODS
Sequencing Analysis
Deep sequencing of baseline plasma samples was performed in 5,397 patients from 22 countries across the HCV Gilead clinical development program from 2010 to 2015. The list of clinical trials and identification numbers are included in the supplement materials (supplemental Table 1). The HCV NS5A coding regions were amplified by DDL Diagnostic Laboratory (Rijswijk, Netherlands) using proprietary amplification primers and standard reverse transcription polymerase chain reaction (RT-PCR) technology, if a plasma sample was available and baseline HCV RNA was >1000 IU/mL. Deep sequencing using MiSeq platform (Illumina, Inc., San Diego, CA) was performed by WuXi AppTec (Shanghai, China) or DDL Diagnostic Laboratory (Rijswijk, Netherlands). Deep sequencing data was split into one file per sample using only 100% matched barcodes to bin the reads. Sequence analysis was performed using internally developed software in a stepwise fashion. Briefly, raw reads from the FASTQ files were trimmed and filtered based on quality scores and read length. Trimming was carried out on reads when quality score decreased below 15, and reads shorter than 50 nucleotides were removed. Deep sequencing data was aligned using MOSAIK v1.1.0017. All aligned reads were then translated in-frame and changes from a reference sequence were determined. Assay sensitivity and assay background cutoffs were evaluated based on plasmid and RNA controls. There are no standardized HCV deep sequencing assays available as commercialized kits, therefore cross-validation of deep sequencing data from DDL and WuXi was performed on a subset of control samples.
Ethics Statement
All studies were conducted in accordance with the Declaration of Helsinki, Good Clinical Practice guidelines, and local regulatory requirements. All patients provided written informed consent.
Definition of NS5A Polymorphisms at RAS Positions (RAPs) and Resistance-Associated Substitutions (RASs)
NS5A RAPs were defined as any change from genotype 1a or 1b reference strains (1a-H77 or 1b-Con1) at NS5A positions associated with NS5A drug resistance. NS5A class RASs were summarized by the HCV Drug Resistance Advisory Group group [14], and/or recently observed in clinical trials with ledipasvir, velpatasvir, daclatasvir, pibrentasvir, and elbasvir [[15], [16], [17], [18], [19], [20], [21], [22], [23]], specifically variants at NS5A positions 24, 28, 30, 31, 32, 38, 58, 92, 93 that confer >2.5-fold reduced susceptibility to any NS5A inhibitor. Ledipasvir-specific RASs were classified as variants at NS5A positions 24, 28, 30, 31, 32, 38, 58, 92, 93 that confer >2.5-100 or >100-fold reduced susceptibility to ledipasvir in vitro or were selected in clinical trials in patients treated with ledipasvir-containing regimens [[2], [24], [25]] (Table 1).
Assessment of Sustained Virologic Response (SVR) in patients with and without pretreatment NS5A Inhibitor RASs
SVR12 rates were assessed only in the 1,765 patients who were treated with currently recommended regimens containing ledipasvir-sofosbuvir (according to AASLD/IDSA and EASL guidelines) in 15 phase 2 and phase 3 Gilead-sponsored clinical trials (supplemental Table 2). Only patients who were not previously exposed to NS5A inhibitors were included in these analyses. Patients were excluded from these analyses if they did not achieve SVR due to non-virologic failure (e.g., lost to follow up). The results were analyzed according to the 1% and 15% detection cut-offs of NS5A RAPs, class RASs or ledipasvir-specific RASs.
RESULTS
Patient Baseline Characteristics
Demographic and baseline clinical characteristics of the 5,393 NS5A inhibitor-naive patients included in the NS5A baseline prevalence RAS analysis are provided in Table 2. The majority of patients were treatment naïve (56%) and male (64%), with HCV genotype 1a (65%) and non-CC interleukin (IL) 28B alleles (73%). Approximately one third (32%) of patients had cirrhosis.
Prevalence and Type of Pretreatment RASs across Geographic Regions
Baseline prevalence of NS5A polymorphisms at RAS positions, NS5A class RASs, ledipasvir RASs and the specific Y93H NS5A variant was evaluated in genotype 1a (n=3501) and 1b (n=1887) patients using 1% through 50% sensitivity cut-offs (Fig. 1). Higher prevalence of all categories for NS5A RASs was observed at 1% sensitivity cut-offs and sharply declined with reduction in sensitivity of variant detection to 15%. No significant changes in NS5A RASs prevalence was observed with further reductions in assay sensitivity from 15% to 50%.
The prevalence of NS5A polymorphisms at RAS positions was significantly higher as compared to NS5A class RASs in both genotype 1a and 1b across all sensitivity cut-offs. The prevalence of NS5A class RASs was about 5% higher than that of LDV RASs in genotype 1a. This difference was mostly represented by prevalence of the M28V NS5A class RAS that is not an LDV RAS. There was little difference between NS5A class and LDV RASs in genotype 1b. Prevalence of Y93H was higher in genotype 1b as compared to genotype 1a across all assay cut offs. Based on the observation of a sharp decline in prevalence from 1% to 15%, further analyses were performed with both 1% and 15% cut-offs.
Overall at the 15% assay cut off, pretreatment NS5A class RASs were detected in 13.0% of genotype 1a patients (Table 3). The prevalence of NS5A class RASs overall in patients with genotype 1a HCV did not differ significantly across most of the geographic regions with the frequency ranging from 12.1% to 15.6%, but the prevalence of ledipasvir RASs was significantly higher among patients in Oceania, than among those from other regions combined (12.7% vs 7.9%, p=0.005). The overall prevalence of baseline ledipasvir RASs in genotype 1a patients was 8.3% with some numeric differences between different regions, the highest in Oceania (12.7%) and the lowest in Europe (7.7%). Specific RASs were detected at a similar frequency in genotype 1a patients across geographic regions, including K24R, M28V/T, Q30H/R, L31M and Y93H.
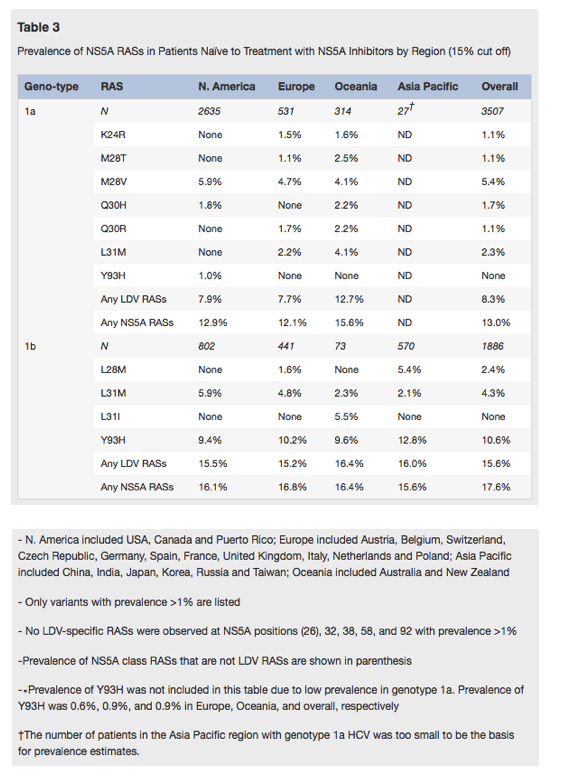
The overall prevalence of baseline NS5A class RASs was slightly higher (17.6%) in patients infected with genotype 1b than in those infected with genotype 1a (Table 3). The frequency of detection of NS5A class RASs ranged from 16.1% to 20.4% in genotype 1b patients with only minor numeric differences across geographic regions. The prevalence of baseline LDV RASs among genotype 1b patients was also similar across different regions (15.2-16.4%). Y93H was detected at a much higher frequency (10.6%) than other RASs, including L28M and L31I/M/V among genotype 1b patients, but differences in the prevalence of each RAS between regions were small. In both subtypes, the prevalence of multiple (⩾2) RASs was low; ranging from 0 to 3.8% in genotype 1a, and all less than 1.5% in genotype 1b.
Assessment of the Effect of Baseline RASs on Treatment Outcome with Ledipasvir-Sofosbuvir by RAS Categories and Sensitivity Cut offs
To evaluate the effect of baseline RASs on treatment outcome, SVR12 rates were assessed in 1,765 patients from 15 ledipasvir-sofosbuvir clinical trials who were treated with currently recommended regimens according to the 2015 AASLD/IDSA and EASL guidelines. The baseline characteristics of this population are given in Table 4. A systematic comparison of the effect on SVR12 rates was performed in genotype 1a and 1b treatment-naive and treatment-experienced patients for NS5A RAPs, class RASs, and LDV RASs, and LDV RASs with >100-fold change, using a 15% sequencing assay cut off (Fig. 2).
Fig. 2
SVR rates in patients with and without NS5A RASs. The figures show the rates of SVR12 by presence at baseline of NS5A polymorphisms at RAS positions (RAPs), NS5A class RASs, and ledipasvir-specific RASs, and ledipasvir-specific RASs that confer >100-fold change at a 15% sensitivity threshold.
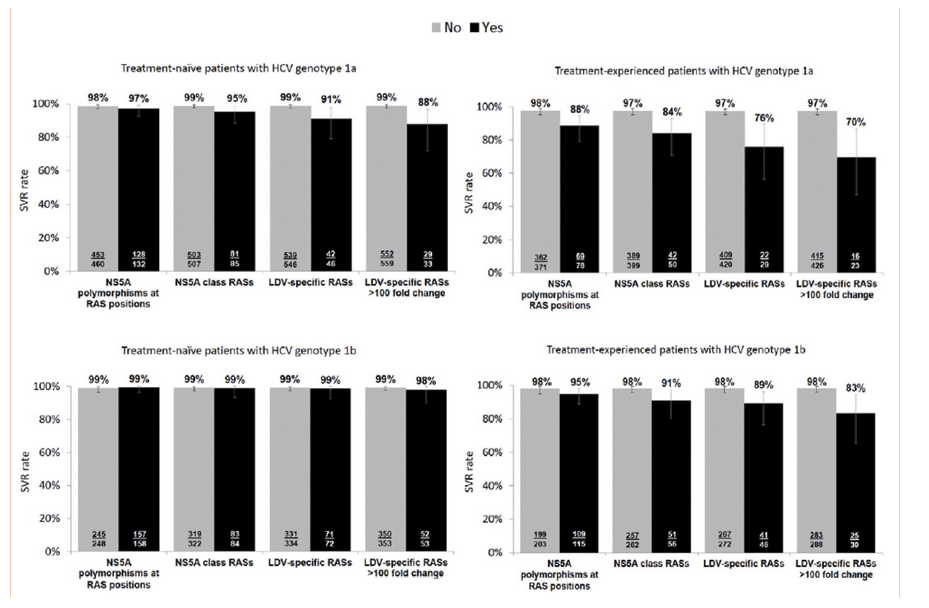
In treatment-naive patients with genotype 1b HCV infection, the presence of baseline NS5A polymorphisms at RAS positions or NS5A class RASs did not impact the treatment outcome with ledipasvir-sofosbuvir regimens with SVR12 rates of 98%-99% in every group. The SVR12 rate in genotype 1a patients with baseline LDV RASs was 94% and 91% (1% and 15% cut offs, respectively) compared to 99% in patients without LDV RASs (Table 5). The presence of baseline LDV RASs in genotype 1b patients had no impact on SVR12 rates.
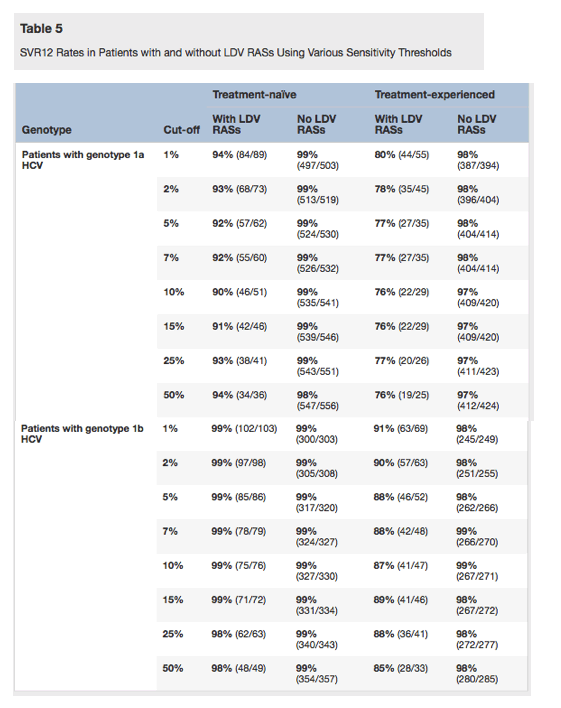
In treatment-experienced patients with genotype 1a or 1b HCV infection, LDV RASs had the most notable impact on SVR12 rates (76%-80% vs 97%-98% and 89%-91% vs. 98% in genotype 1a and 1b, respectively). Even though similar results were obtained when 1% and 15% sensitivity assay cut offs were used, SVR12 rates were slightly lower when 15% assay cut off was used.
Taken together, the comparison of the different categories of NS5A RASs and assay cut offs, LDV RASs detected with a 15% assay cut off was identified as the most discriminating for ledipasvir-sofosbuvir regimen baseline analyses and this cut off was used to perform further subgroup evaluations.
Effect of Baseline Ledipasvir RASs on Treatment Outcome by Patient Population
SVR12 rates by treatment history and cirrhosis status according to baseline ledipasvir RASs using a 15% assay cutoff was performed for HCV genotype 1a and genotype 1b (Fig. 3) infected patients. The SVR12 rate in treatment-naïve non-cirrhotic patients was not substantially impacted by the presence of ledipasvir RASs at baseline (92% SVR). Numerically lower SVR12 rates (86%) were observed in treatment-naïve cirrhotic genotype 1a patients with baseline ledipasvir RASs but only 7 patients limits the interpretability of this finding. Of genotype 1a patients, prior exposure to HCV treatment appeared to impact the SVR12 rates in both non-cirrhotic and cirrhotic groups (75% and 77% respectively) in the presence of baseline ledipasvir RASs, but the number of patients in these groups was also small (<20). Of genotype 1b patients, the SVR12 rates remained >90% across the groups regardless of treatment history and presence of cirrhosis with or without baseline ledipasvir RASs, except for the treatment-experienced non-cirrhotic group which showed an SVR rate of 87%, but it only included 23 patients. The number and prevalence of patients with multiple RASs were small (N <30, <1%) for both ledipasvir-specific and NS5A class RASs in genotype 1a and 1b. The overall SVR rates were 64% (9/14) in genotype 1a and 100% (2/2) in genotype 1b patients with multiple ledipasvir RASs (supplemental Table 3). Of those with multiple NS5A class RASs, the SVR rates were 74% (17/23) and 83% (5/6) among genotype 1a and 1b, respectively (supplemental Table 3).
Fig. 3
SVR12 rates by treatment history and cirrhosis status. The figures show the rates of SVR12 by regimen, treatment history (naïve vs previously treated), and cirrhosis status (present vs absent). NC = non-cirrhotic, C = cirrhotic, TE = treatment-experienced, and TN = treatment-naïve.
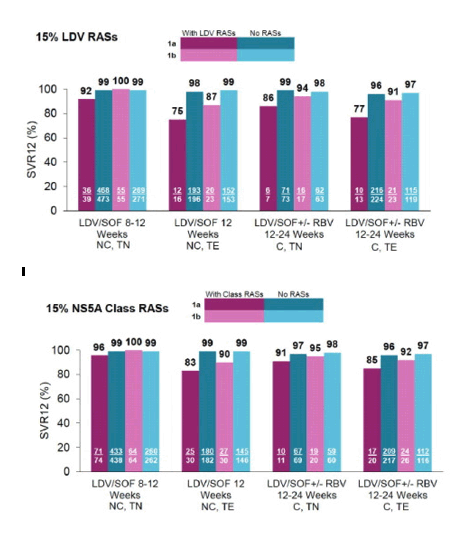
Among patients with cirrhosis, there were too few patients with baseline RASs to further assess the impact of treatment duration and/or the addition of ribavirin on treatment outcome (supplemental Table 4).
DISCUSSION
Current NS5A inhibitors show overlapping but distinct resistance profiles with RASs described at the NS5A amino acid positions 24, 28, 30, 32, 31, 38, 58, 92, and 93. There are advantages and disadvantages with each of the three main approaches to defining NS5A RASs. The advantage of using the NS5A RAPs definition is that it provides a uniform list of variants for all NS5A inhibitors. It does not require extensive phenotypic testing of all variants with several NS5A inhibitors and provides inclusive assessment of variants that developed in patients treated with NS5A inhibitors. However, for baseline analyses that investigate the role of pre-existing variants on treatment outcome, substitutions that are fully susceptible to a specific NS5A inhibitor dilute the investigated effect. To characterize NS5A class RASs, i.e. those that show reduced susceptibility to one or more NS5A inhibitors in vitro, standardized phenotypic testing is needed for each NS5A inhibitor. Even though the NS5A class RAS definition would exclude variants that are known to be sensitive to NS5A inhibitors and thus provide a more sensitive analysis of the effect of baseline RASs on SVR, some attenuation of the signal may still be observed due to different resistance profiles among the NS5A inhibitors. With further optimization of NS5A inhibitors to improve resistance profiles, the list of NS5A variants and positions that confer reduced susceptibility to the next generation drugs is shortening. Using drug-specific RASs is the most scientifically rigorous way to perform efficacy and baseline resistance analyses. However, extensive standardized phenotypic testing is needed to accurately define drug-specific RASs. Additionally, novel resistance substitutions that develop rarely in vivo may be missed during resistance monitoring and it may be difficult to compare results to those from other studies since drug-specific RASs will be different between various NS5A inhibitors. Another disadvantage of using drug-specific RASs is that this definition fails to capture relevant information regarding the response in patients with resistance to other NS5A inhibitors.
The results presented here show that analysis of ledipasvir drug-specific RASs shows more impact on ledipasvir-sofosbuvir treatment outcomes overall as compared to the analysis of RAPs or class RASs, as would be predicted based on these RASs having demonstrated reductions in susceptibility to ledipasvir. However the presence of drug-specific RASs may affect SVR12 rates to a greater or lesser extent depending on the specific pharmacology of an inhibitor and the drug combination regimen being utilized for treatment. For example, previous analyses of ledipasvir-sofosbuvir clinical trials have shown that only ledipasvir-specific RASs contributing >100-fold reduction in susceptibility result in lower SVR rates with ledipasvir-sofosbuvir regimens [2].
As multiple options for HCV treatment containing NS5A inhibitors have become available and more broadly applicable, understanding the prevalence of baseline NS5A RASs in specific regions has become more important. In this comprehensive analysis using >5,000 patient samples from 21 countries in 4 continents, it is shown that the prevalence of both NS5A class and ledipasvir RASs does not differ significantly across regions for both genotype 1a and 1b. Numerically lower prevalence of NS5A RASs is observed for genotype 1a in Asia Pacific, but there were small numbers of genotype 1a patients included from this region (n=27) for epidemiological reasons. The prevalence of specific NS5A class and ledipasvir RASs is also similar across regions for both genotypes 1a and 1b. For genotype 1b, the prevalence of Y93H was the highest in Asia Pacific whereas the prevalence of L31M/I/V was the lowest in this region. It must be noted, however, that large regions of the world-including much of Asia, and all of Africa, South America and the Caribbean-are not represented in this analysis.
The rates of SVR among patients without pretreatment ledipasvir RASs at all detections thresholds were high regardless of subtype and treatment history, ranging from 97% to 99%. The greatest impact of ledipasvir RASs on SVR was among treatment-experienced patients with genotype 1a HCV, who had an SVR rate of 76% (at the 15% cut-off). This difference was approximately the same at all detection thresholds. Among treatment-naïve patients with genotype 1b HCV, pretreatment ledipasvir RASs appeared to have little to no impact on SVR, with rates ranging from 98-99% for all detection thresholds. Treatment-naïve patients with genotype 1a HCV and treatment-experienced patients with genotype 1b HCV fell somewhere in between, with differences of 4% to 10% between those with and without ledipasvir RASs.
The clinical interpretation of these findings remains challenging. The decision to perform pre-treatment RAS testing may be made based on the magnitude of the effect of these RASs on treatment outcome. The effect of NS5A or ledipasvir-specific RASs on treatment outcome was greatest in treatment-experienced patients and/or those with cirrhosis, groups that are at highest risk of disease progression. An argument in favor of pre-treatment RAS testing could thus be made, with the decision to possibly extend treatment duration and/or add ribavirin for those with ledipasvir-specific RASs. However, it should be noted that the number of patients within these subgroups was small (⩽23 patients) and these data may not be generalizable to the broader population.
|
|
| |
| |
|
|
|