| |
Cure Research Review at OAR NIH Meeting
|
| |
| |
Download the PDF here
Download the PDF here
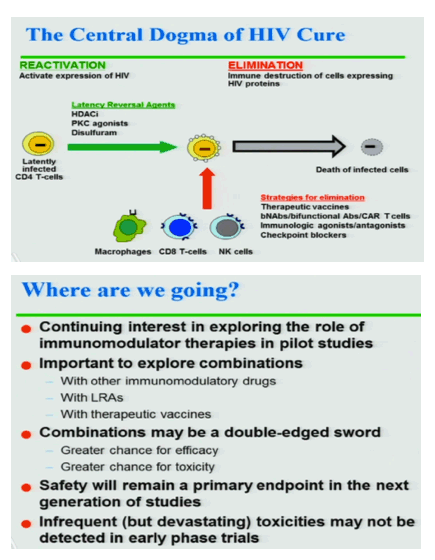
Clinical Trials of HIV Cure: Where have we been? Where are we going? talk by Dan Kuriztkes
At around 50 minutesDan Kuritzkesbegins his interesting review & summary on Cure Research at the OAR ARAC recent meeting just following the 3 day Cure research NIH meeting in Maryland, and full ARAC panel discussion of Cure research follows Dan's talk
https://videocast.nih.gov/summary.asp?Live=20129&bhcp=1
neutralizing antibodies may be more effective in suppressing HIV as therapy perhaps as maintenance as long acting therapy - rather than alone or in combination to reactivate HIV & be effective for cure. It also remains unclear how NAs would be effective in suppressing HIV. What has been the subject of cure research recently has been latency reactivating agents, "at current dosing regimens, LRAs are only modestly effective in reactivating latent HIV-1.... reactivating the entire reservoir will be challenging. It is highly unlikely that reactivation alone will suffice to deplete the latent reservoir of HIV-1. Safety & infrequent toxicities potentials that may not be detected in early phase studies will be focus of research. Safety oflatency reactivating agents is questioned by some, they are used in cancer therapy & can have unknown long term concerns because they up and down regulate gene expression but it is not clear that ongoing LRA cure research studies adequately warn patient study participants of these risks. Safety concerns of new checkpoint inhibitors used for cancer has recently come to light more so the risk/benefit ratio is questionable. There has been quite a bit of recent discussion about the inadequacy of "informed consent" of patient study participants with recent discussions that participants usually do not appear to fully understand the potential risks of these studies nor of treatment interruption which results in repopulating reservoirs with HIV after years of fully suppressive HAART.
New Study Announced several weeks ago will study TLR9 + 2 neutralizing antibodies - 10-1074 & 3BNC117
New HIV Eradication Combo Study - (01/13/17)
HIV-1 antibody 3BNC117 suppresses viral rebound in humans during treatment interruption - (06/27/16)
Gilead Awards More Than $22 Million in Grants to Support HIV Cure Research - (01/13/17)
TLR9, TLR7 HIV Cure Research - (01/24/17)
-----------------------
other related studies
Toll-like Receptor 9 Agonist Treatment in Chronic HIV-1 Infection (TEACH)
https://clinicaltrials.gov/ct2/show/NCT02443935?term=tlr9&rank=1
Clinical Trial: 3BNC117 and 10-1074 in HIV Uninfected Adults
https://clinicaltrials.gov/ct2/show/NCT02824536?term=10-1074&rank=1
A Study of the Safety, Pharmacokinetics and Antiretroviral Activity of 10-1074 https://clinicaltrials.gov/ct2/show/NCT02511990?term=10-1074&rank=2
-------------------------
Broadly Neutralizing HIV-1 Antibodies (bNAbs) are neutralizing antibodies which neutralize multiple HIV-1 viral strains.[1] In contrast, non-bNAbs are specific for individual viral strains. The discovery of bNAbs has led to an important area of research, namely, discovery of a vaccine, not only limited to HIV, but also other rapidly mutating viruses like Influenza, etc.
https://en.wikipedia.org/wiki/Broadly_neutralizing_HIV-1_antibodies
-------------------------
Antibody 10-1074 suppresses viremia in HIV-1-infected individuals
Nature Medicine Jan 2017 Marina Caskey
ABSTRACT: Monoclonal antibody 10-1074 targets the V3 glycan supersite on the HIV-1 envelope (Env) protein. It is among the most potent anti-HIV-1 neutralizing antibodies isolated so far. Here we report on its safety and activity in 33 individuals who received a single intravenous infusion of the antibody. 10-1074 was well tolerated and had a half-life of 24.0 d in participants without HIV-1 infection and 12.8 d in individuals with HIV-1 infection. Thirteen individuals with viremia received the highest dose of 30 mg/kg 10-1074. Eleven of these participants were 10-1074-sensitive and showed a rapid decline in viremia by a mean of 1.52 log10copies/ml. Virologic analysis revealed the emergence of multiple independent 10-1074-resistant viruses in the first weeks after infusion. Emerging escape variants were generally resistant to the related V3-specific antibody PGT121, but remained sensitive to antibodies targeting nonoverlapping epitopes, such as the anti-CD4-binding-site antibodies 3BNC117 and VRC01. The results demonstrate the safety and activity of 10-1074 in humans and support the idea that antibodies targeting the V3 glycan supersite might be useful for the treatment and prevention of HIV-1 infection
--------------------------
Elimination of HIV-1-infected cells by broadly neutralizing antibodies
http://www.nature.com/articles/ncomms10844
Our results directly demonstrate that bNAbs, in addition to other immune interventions43,44, represent an efficient tool for consideration in 'shock and kill' strategies aimed at purging the viral reservoir14,26. A bNAb-based HIV-1 cure will likely require a personalized screening of the pattern of Env epitope exposure on reactivated cells, to determine the optimal combination of antibodies. The reported data on differences among bNAbs in their ability to kill HIV-1-infected lymphocytes will enable a better understanding of the functional attributes of antibodies for prevention and cure strategies.
We have analysed here the ADCC activity of bNAbs against laboratory-adapted, transmitted/founder and reactivated HIV-1 derived from the viral reservoir. We report that a subset of bNAbs effectively kills HIV-1-infected lymphocytes. All antibodies tested contain the same Fc region, implying that the differences in their ability to signal through FcγRIII and mediate ADCC is dependent on their variable regions. Thus, differences in killing efficiency are likely due to changes in binding of the antibody and accessibility of the Fc region when bound to infected cells. Env steady-state levels and surface stability of bNAbs regulate their ADDC potency. We show a significant correlation between, the intensity of antibody binding at the cell surface, the stability of this binding at 37 °C, neutralization activity and their capacity to eliminate HIV-1-infected cells. By using the related antibodies 10-1074 and 10-1369, we further demonstrate a link between the affinity of a given antibody to the Env trimer and its ADCC potency. Our results extend previous work showing that antibody affinity is related to neutralizing activity36 and provide a comprehensive mechanistic analysis of the ADCC activity of a panel of the newest bNAbs. We show that Env epitope exposure on infected cells is highly variable, depending on the viral isolate. Future work will help assess the role of viral proteins and other factors in the modulation of epitope exposure and ADCC. PHA-activated CD4+ T lymphocytes derived from patients' cells expose sufficient amounts of Env epitopes required for recognition by combinations of bNAbs and killing by NK cells. Our experiments offer a mechanistic explanation as to how bNAbs associated with viral inducers decrease rebound from latent reservoirs in humanized mice27. We further show that induced viral reservoirs display an extreme heterogeneity in Env epitope exposure. This reflects the variable sensitivity of virions from the reservoir to neutralization by bNAbs37. It would be worthwhile to follow the landscape of epitopes longitudinally, in order to determine whether reactivated founder viruses evolve over time, and originates from clones or populations of cells38,39 with homogeneous or heterogeneous Env profiles. In HIV-1-infected individuals, low-level viraemia during effective highly active antiretroviral therapy likely result from expression of archival virus and covert viral replication40. Latent proviruses are found predominantly in subsets of resting memory cells, which are largely non-permissive for viral gene expression35,41,42. It will be of interest determining whether bNAbs may kill with similar efficiencies cells from the latent reservoir, or from a population of lymphocytes with ongoing low-level of viral replication. This may be achieved by sorting resting memory T cells from patients and performing ADCC assays with various cell activators or latency-reversing agents35. It will also be of interest to assess whether the bNAbs may kill the actual reactivated latently infected cells before the virus spreads to neighbouring cells. This could be performed by visualizing killing of infected cells after viral reactivation in humanized mouse models.
---------------------------
Broadly Neutralizing Antibodies for HIV-1 Prevention or Immunotherapy
Marina Caskey, M.D., Florian Klein, M.D., and Michel C. Nussenzweig, M.D., Ph.D.
N Engl J Med 2016
Despite tremendous efforts to prevent transmission of human immunodeficiency virus type 1 (HIV-1), new infections and AIDS-related deaths continue to be a global problem. Antiretroviral therapy (ART) can effectively suppress HIV-1 replication and limit disease progression. However, ART fails to eradicate the virus, and suppression requires lifelong therapy, which may have side effects and poses a risk of the development of resistance. New approaches to preventing and treating HIV-1 infection are therefore necessary to contain the epidemic and to bolster nascent efforts to find a cure.,
A new generation of highly potent broadly neutralizing antibodies (bNAbs) may represent a promising approach to combating HIV-1 infection.1 Although antibodies that neutralize HIV-1 were discovered shortly after the disease was first described, most HIV-1-infected people produce nonneutralizing antibodies or antibodies that neutralize only a limited number of different HIV-1 strains. However, high serum titers of HIV-1 neutralizing activity develop in a small fraction of infected persons. In contrast to the typical antibody response, bNAbs can take years to develop, and it has not yet proved possible to elicit them by standard immunization strategies.
Isolation of bNAbs from infected persons with high levels of HIV-1-neutralizing serum activity (so-called elite neutralizers) was not easily achieved until the advent of single-cell-based anti-HIV-1 antibody cloning techniques.2 The anti-HIV-1 antibodies obtained by this and subsequently developed related methods had a number of unusual characteristics that partially account for the difficulty of developing a vaccine that can elicit bNAbs. The most prominent of these features is a high level of somatic hypermutation, which is required to accommodate binding to the highly glycosylated viral envelope protein. Some of the new bNAbs can neutralize up to 90% of HIV-1 strains worldwide at low concentrations.1 Moreover, bNAbs recognize many sites on the viral envelope spike. Antibodies currently being tested or under development for clinical use target the CD4-binding site, a glycan patch surrounding the V3 loop, and the apex of the HIV-1 spike.3 It thus seems possible that they could be used in combination to prevent or control HIV-1 infection. As with other drugs, the efficacy of any single anti-HIV-1 antibody in preclinical and clinical trials is related to the potency and breadth of its activity against different HIV-1 isolates. Like monotherapy with small-molecule antiretroviral (ARV) drugs, the administration of a single anti-idiotypic bNAb selects for resistant variants. As recently reported,3 in work that Bar et al. have extended, bNAbs are being studied as a potential way to suppress viremia during analytical treatment interruption. The degree of efficacy in maintaining viral suppression in these and other studies was most likely related to the potency of the bNAbs.4,5
However, antibodies are currently expensive to produce and must be administered parenterally. In contrast, small-molecule ARV drugs can be synthesized at low cost, are administered orally, and are highly effective. So it is important to ask why we should bother investigating the use of antibodies for HIV-1 prevention or therapy.
Several unique features of antibodies make them interesting to explore as drugs for HIV-1 prevention and therapy. First, antibodies differ from small-molecule drugs in having naturally long half-lives of 2 to 3 weeks. It was this property that made it possible, before the advent of an active hepatitis vaccine, to administer pooled antihepatitis immunoglobulins as a passive vaccine to prevent infection in travelers for up to 3 months. The same is true for bNAbs and HIV-1 prevention. A single administration of anti-HIV-1 bNAbs can protect macaques for up to 23 sequential weekly challenges.5 Moreover, the antibody half-life can be increased by a factor of 2 to 4 by point mutations in the constant (Fc) region of the molecule. For example, in clinical investigations, an anti-respiratory syncytial virus antibody that carries half-life extension mutations showed neutralizing activity for 240 days after administration in humans. When similar mutations were introduced into anti-HIV-1 bNAbs, they resulted in a prolonged half-life and extended the protective effects in macaques.5
The pharmacokinetic properties of anti-HIV1 bNAbs with half-life-extending mutations are currently being tested in humans. It is entirely possible that bNAbs could be used as passive vaccines administered subcutaneously on a quarterly or a biannual basis. In contrast, the best small-molecule drugs in late-stage clinical testing have to be administered intramuscularly every 2 months for prevention or therapy. More important, decaying levels of these long-acting drugs may allow selection of resistant viral strains, and persons in whom oral or injectable small-molecule drugs fail to prevent infection because of breakthrough resistant viruses will probably have no response to antiretroviral regimens containing the same drug classes. In contrast, infections with breakthrough antibody-resistant strains would be very likely to be sensitive to standard ARVs, since the targets of the bNAbs that are currently in clinical testing do not overlap with those of approved ARVs.
Another reason to explore bNAbs in the context of HIV-1 infection is their potential as immunotherapeutics. This idea is similar to the rationale for using antibodies in cancer immunotherapy: antibodies engage the host immune system to fight disease. They do so by binding the target cell, which is then flagged for killing and phagocytosis by host leukocytes. One of the results of this type of immune opsonization is enhanced target-cell clearance. A second effect is to activate dendritic cells by opsonized immune complexes, leading to enhanced antigen processing and presentation to T cells. Activated T cells can kill target cells directly or act as helper cells for antibody responses. In the context of cancer therapy, host immune responses can produce lasting remissions. In the HIV-1 context, they can control viral replication in the absence of drugs, as seen in some HIV-1-infected patients.
The potential contribution of immunotherapy in the context of HIV-1 infection may be particularly important when it comes to the possibility of HIV-1 eradication. In phase 1 clinical trials, bNAbs enhanced clearance of HIV-1, infected cells, or both and boosted host humoral immunity to HIV-1.
These antibody-mediated immune-enhancing effects might lead to better control of viral replication and reduce the size of the long-lived latent reservoir that persists despite suppressive ART. It is important to note, however, that the immune-engaging features of antibodies are mediated through binding of antibody to available antigen. Suppressive ART may therefore dampen these potential effects by a nearly complete shutdown of viral replication. It will therefore be important to further evaluate the effects of bNAbs in combination with strategies aimed at reactivating latent proviruses in the presence or absence of standard ART.
Although bNAbs have attractive properties, particularly for HIV-1 eradication strategies, as compared with standard ART, resistance does develop during antibody monotherapy. Moreover, preexisting resistance to individual bNAbs, as described in our work3,4 and by Bar and colleagues, may also pose a challenge to their use, especially during chronic HIV-1 infection. We speculate that a combination of bNAbs, like combinations of small-molecule ARV drugs, can avert selection of escape variants. Ongoing studies aim to explore their use for HIV-1 prevention and immunotherapy in infected persons.
|
|
| |
| |
|
|
|