| |
PrEP: State of the Science (ASM/ICAAC June 2017)
|
| |
| |
Kenneth H. Mayer, MD
Reported by Jules Levin
ASM Microbe 2017 Meeting
June 4th, 2017
- Persons likely to benefit from using PrEP
- Safety, Adherence
- PrEP if serodiscordant partner has a suppressed viral load? PARTNER Study
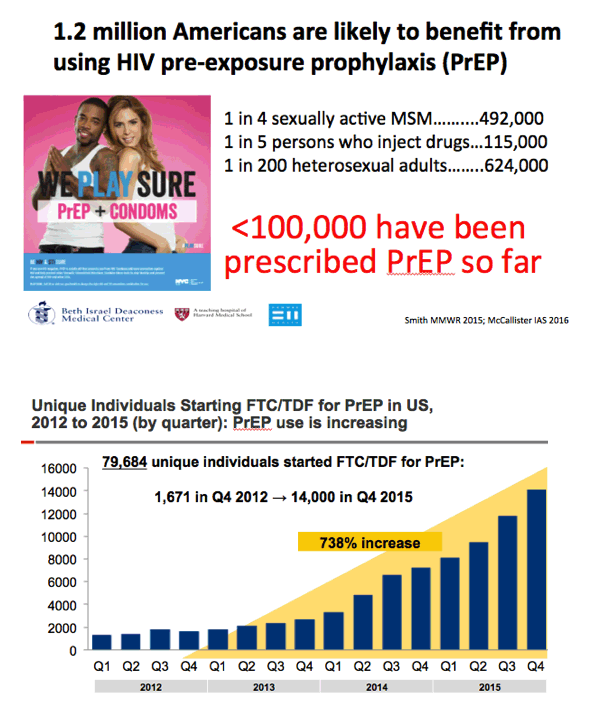
- 1.2 million Americans are likely to benefit from using HIV pre-exposure prophylaxis (PrEP)
-Penetration of Tenofovir in Mucosal Tissues
- Case Reports: HIV Infection with High Adherence to PrEP
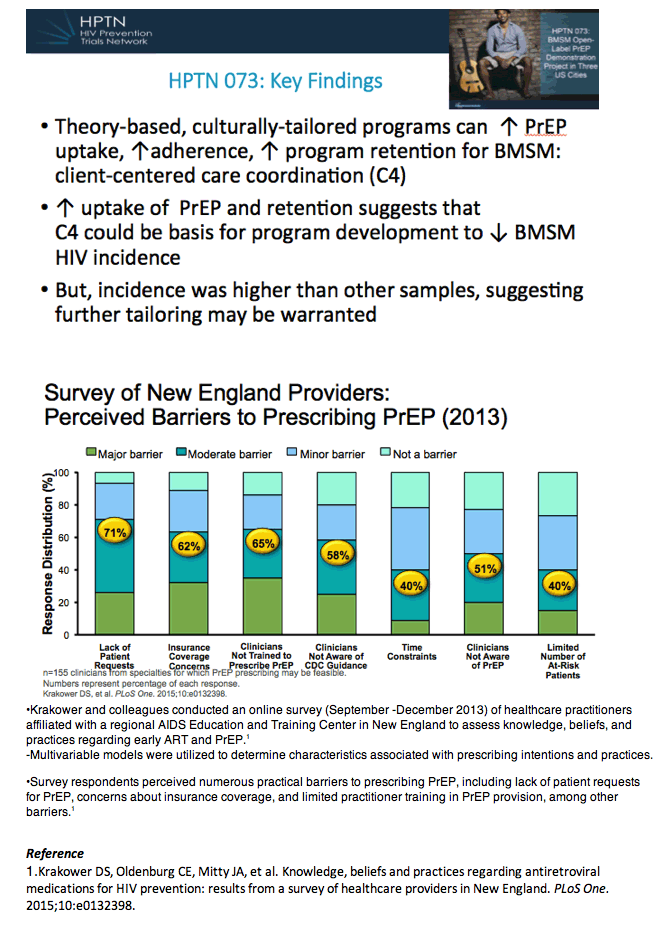
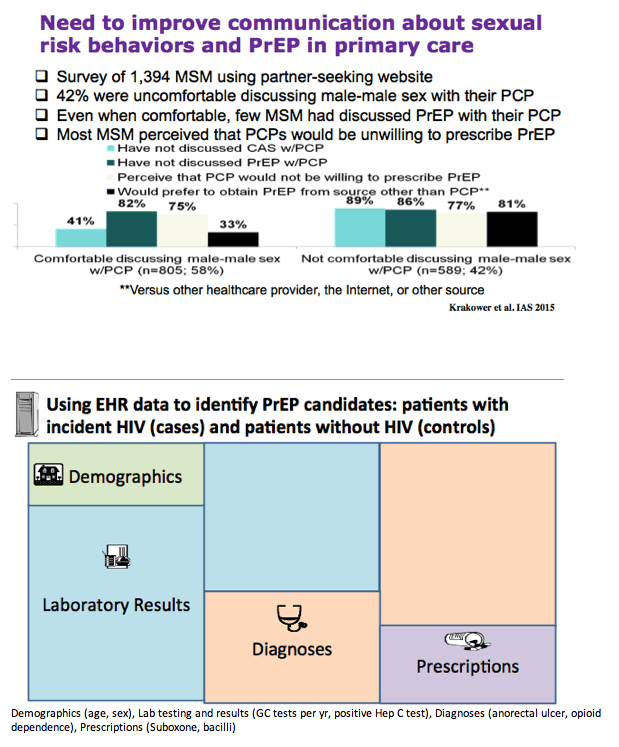
-Need to improve communication about sexual risk behaviors and PrEP in primary care
-PrEP Utilization by Gender and Race in the US
- Unique Individuals Starting FTC/TDF for PrEP in US,
- 2012 to 2015 (by quarter): PrEP use is increasing
-STIs
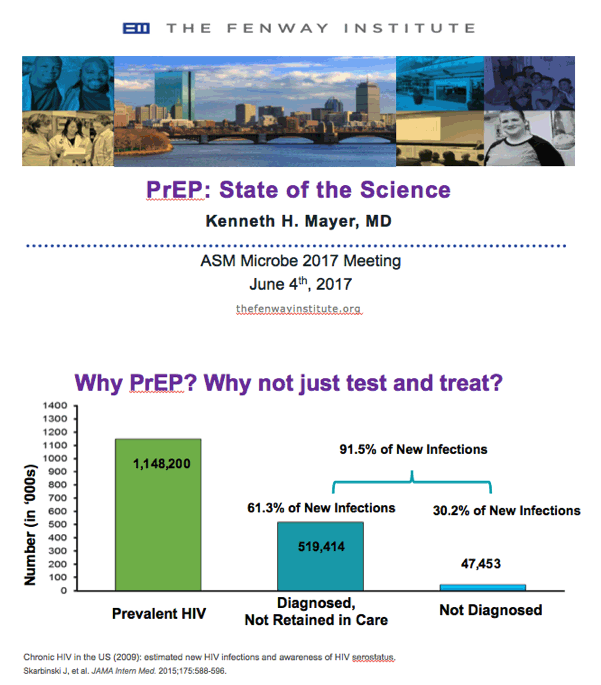
⋅Skarbinski and colleagues conducted a multistep, static, deterministic model that combined population denominator data from the National HIV Surveillance System with detailed clinical and behavioral data from the National HIV Behavioral Surveillance System and the Medical Monitoring Project to estimate the rate and number of transmissions along the care continuum. This analysis reflects the HIV-infected population in the United States in 2009.1
⋅Persons who are HIV infected but undiagnosed and persons diagnosed as having HIV but not retained in care accounted for 91.5% (30.2% and 61.3%, respectively) of the HIV transmissions estimated to have occurred in the United States in 2009.1
Reference
Skarbinski J, Rosenberg E, Paz-Bailey G, et al. Human immunodeficiency virus transmission at each step of the care continuum in the United States. JAMA Intern Med. 2015;175:588-596
FTC, emtricitabine; PrEP, pre-exposure prophylaxis; TDF, tenofovir disoproxil fumarate.
References
1. Marrazzo JM, et al. N Engl J Med. 2015;372:509-518.
2. Van Damme L, et al. N Engl J Med. 2012;367:411-422.
3. Grant RM, et al. N Engl J Med. 2010;363:2587-2599.
4. Thigpen MC, et al. N Engl J Med. 2012;367:423-434.
5. Baeten JM, et al. N Engl J Med. 2012;367:399-410.
6. McCormack S, et al. Lancet. 2016;387:53-60.
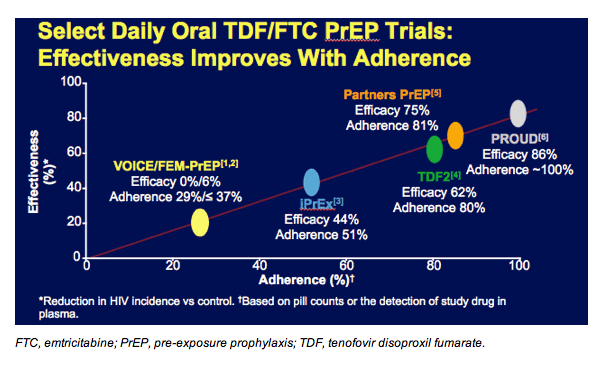
Overall, these randomized clinical trials of PrEP using TDF/emtricitabine have shown that HIV incidence decreases with patient adherence. The y axis depicts the effectiveness of HIV protection in each trial while the x axis documents each trial’s adherence level. Adherence was based on pill counts or the detection of study drug in plasma.
A very clear and strong linear relationship exists where greater adherence across the study population tracks with greater HIV protection. This emphasizes how important it is to be adherent to PrEP. The TDF2, Partners PrEP, and PROUD studies all demonstrated very high degrees of HIV protection in populations that took PrEP consistently.

AE, adverse event; BMD, bone mineral density; FTC, emtricitabine; MSM, men who have sex with men; PBO, placebo; PrEP, pre-exposure prophylaxis; TDF, tenofovir disoproxil fumarate; TGW, transgender women.
A meta-analysis of data from randomized, placebo-controlled PrEP trials demonstrated that the risk of any adverse event or of high-grade adverse events was no different for TDF-based PrEP compared to placebo. Thus, PrEP did not increase the risk of adverse outcomes overall.
Several trials have assessed bone safety during PrEP treatment, including an iPrEx substudy. In high-risk men and transgender women who have sex with men who received daily TDF/FTC PrEP, there was a small net decrease in spine and total hip bone mineral density (BMD) vs placebo at 24 weeks. However, there was no difference in the fracture rate between the groups, and BMD loss that was associated with PrEP recovered once PrEP was discontinued.
As previously discussed, PrEP users are expected to discontinue. In general, PrEP is not expected to be a lifelong medication. So while there appear to be changes in bone mineral density, they have not been clinically relevant, and bone mineral density is recovered when PrEP is discontinued.
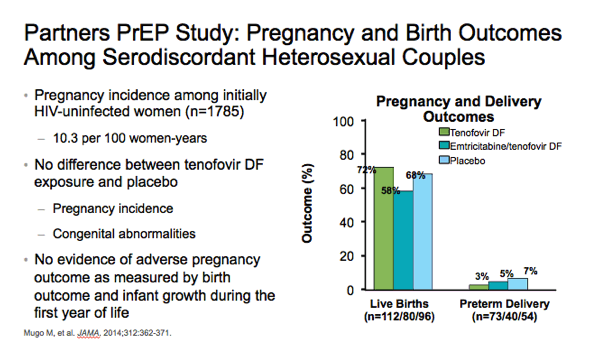
⋅As part of the Partners PrEP study, Mugo and colleagues evaluated pregnancy incidence and birth outcomes among African women. The pregnancy incidence among initially HIV-uninfected women (n=1785) was 10.3 per 100 women-years. There was no difference between the tenofovir DF and placebo arms with regard to pregnancy incidence, live births, preterm deliveries, and congenital abnormalities.1
⋅In addition, there was no evidence of adverse pregnancy outcome as measured by birth outcome and infant growth during the first year of life.1
Reference
1.Mugo NR, Hong T, Celum C, et al. Pregnancy incidence and outcomes among women receiving preexposure prophylaxis for HIV prevention: a randomized clinical trial. JAMA. 2014;312:362-371.

Fem-PrEP: In seroconverters: 26% had drug detected at beginning of infxn window, 21% at end, 15% at both; In non-seroconverters: 25% at beginning, 37% at end, 24% at both (in comparison to 80% in non-seroconverters in TDF2 and partners PrEP)

One concern for women is that tenofovir does not penetrate as well into cervicovaginal tissue as rectal tissue. So, these are data looking at detection of the medication after a single dose of TDF/FTC. And particularly on the right-hand side, are tenofovir diphosphate levels in the different compartments, which you can see was detected in rectal tissue all the way through 14 days and was detected in vaginal and cervical tissue during that period of time. But tenofovir itself in the blood—assuming you have free tenofovir—in the cervical and vaginal tissue was not detected for as long a period of time. So, these data might suggest that women because of their mode of exposure may have to be particularly adherent to these medications in order to get maximal benefit.
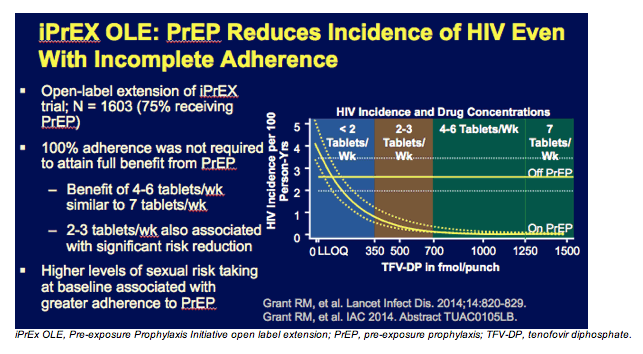
This slide shows a post-hoc analysis from iPrEx, which suggests that among individuals whose blood levels were consistent with taking at least 4 tablets a week of the TDF/FTC regimen, there were no new HIV infections seen in that population. This doesn't prove that 4 tablets a week are as good as 7 tablets a week, but it certainly suggests that at least for MSM, there may be some forgiveness if a dose is missed periodically.
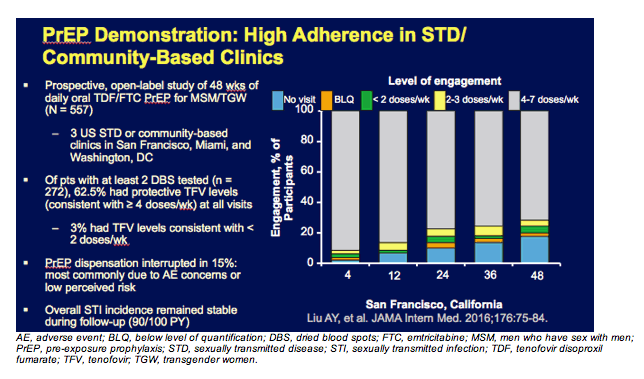
In this slide, you can see PrEP rolling out in the real world. These are data from a demonstration project that was conducted in San Francisco and in Washington, DC and in Miami. In San Francisco and Miami, the sites were STD clinics, and in Washington, it was at an LGBT health center. And you can see over the course of 48 weeks—almost a year—the vast majority of individuals still had drug levels consistent with taking at least 4 pills a week, so a pretty high level of adherence maintained over the course of a year without any of the further additional supports that are common in a clinical trial. These were just individuals getting access to PrEP and then being asked to come in quarterly for followup visits.
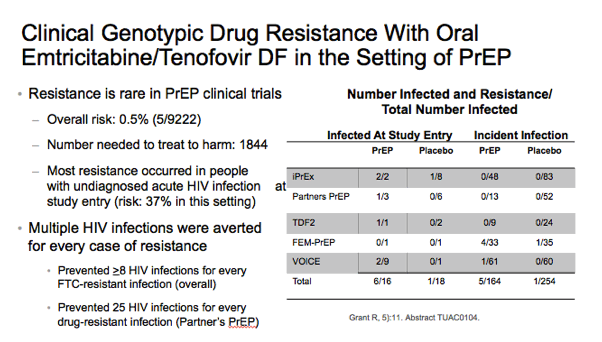
⋅The emergence of resistance is rare in PrEP clinical trials (0.05% of exposed, 5/9222). Most cases occurred in people with undiagnosed acute HIV infection at study entry. To keep these numbers in perspective, multiple HIV infections were averted for every case of resistance. For example, in the Partners PrEP trials, 25 HIV infections were prevented for every drug-resistant infection caused. PrEP-selected resistance in plasma decays below detection by 6 months following drug cessation and remains undetectable for at least 24 months.1
Reference
1.Grant R, Fonner V, Rodolph M, et al. Benefits of pre-exposure prophylaxis relative to drug resistance risk. JAIDS. 2016;19(suppl 5):11. Abstract TUAC0104.

However, as illustrated by this case reported at CROI 2016, PrEP is never going to necessarily be 100% effective. This was a MSM from Toronto, Canada. His clinician had a strong index of suspicion when the patient reported substantial risks for HIV and came in with some atypical symptoms. So, the clinician ordered an acute HIV infection assessment, did an RNA testing, and at the same time saved blood to test for TDF/FTC and found that the patient, indeed, was highly adherent to the study medication.
When the RNA test came back positive, the clinician ordered resistance testing, which suggested a high level of resistance, including resistance to some of the drugs that are not contained in a PrEP regimen of TDF/FTC. For example, the patient had resistance to integrase strand inhibitors and nonnucleoside reverse transcriptase agents and protease inhibitors, suggesting that this is a case of a patient having a highly resistant strain of HIV and transmitting it despite PrEP. So, this is a case in point that makes us say that we cannot guarantee PrEP being 100% effective, but it's certainly highly effective. So, if individuals really want maximal protection, there's still a rationale for using condoms. The other rationale for using condoms even in the setting of using PrEP is to avoid other sexually transmitted infections, because PrEP only protects against HIV.
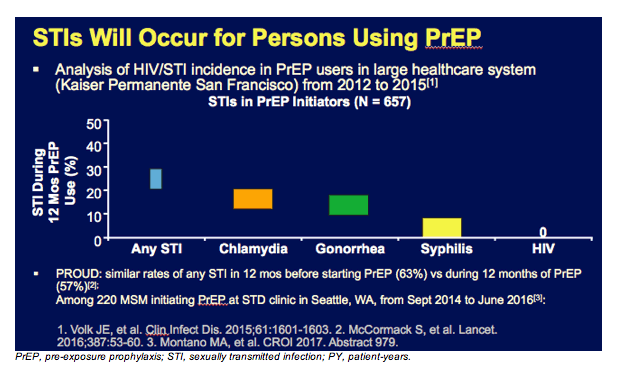
Sexually transmitted infections will occur in persons who are receiving PrEP. That is why STI screening at least every 6 months, or potentially more often by clinical discretion, is recommended for PrEP recipients.
The graph on this slide derives from a large healthcare system, Kaiser Permanente San Francisco, where STI rates were measured in about 700 MSM who had initiated PrEP. In the first year of treatment, 50% of individuals developed an STI. In total, 33% had chlamydia, 28% had gonorrhea, and 6% had syphilis, but there were no HIV infections. The MSM in this population likely demonstrated high rates of STI acquisition even prior to PrEP use. This is certainly true for individuals enrolled in the PROUD study where HIV acquisition was reduced by 86% with immediate PrEP use. That population had an STI rate of 63% in the 12 months before starting PrEP and 57% in the 12 months after starting PrEP, showing no new increase in STI rates with PrEP, but really high STI rates overall. This emphasizes that for populations who choose PrEP, STI rates will be high. This is also where PrEP is most needed.
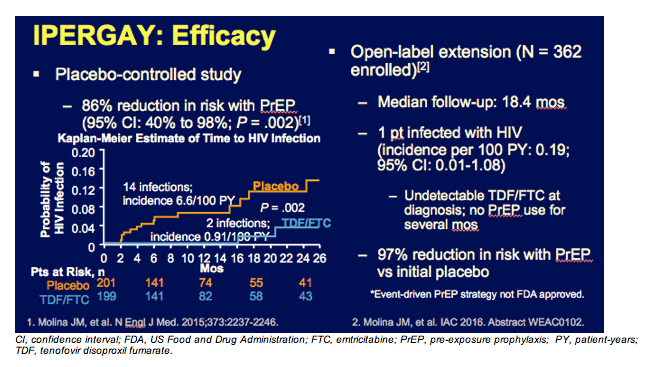
You can see the efficacy data from the IPERGAY study on this slide, with an impressive 86% reduction in risk. And to reiterate, although further discussion about less-than-daily PrEP is under way, the official recommendation is daily usage of PrEP.
The IPERGAY open-label extension showed that only 1 subsequent patient became infected with HIV. This individual did not have any TFV or FTC in detectable in his blood when he became infected. The overall protection rate for event-driven PrEP in this population was 97%.

Joel E. Gallant, MD, MPH:
IPERGAY was a randomized double-blind PrEP study of event-driven oral emtricitabine (FTC)/tenofovir disoproxil fumarate (TDF) vs placebo in high-risk French and Canadian men who have sex with men (MSM).[1] (Note that this intermittent dosing schedule is not approved in the United States.) Although on-demand FTC/TDF reduced the risk of HIV infection by 86%, the incidence of STIs was high in both arms of the study. Data from a clinic in Seattle, Washington, also found a high incidence of chlamydia and gonorrhea in 220 PrEP users.[2] However, the question of whether the use of PrEP actually increases the risk of STIs remains controversial and unanswered.


When should PEP be transitioned to PrEP? So PEP is a response to an acute exposure. If individuals are at recurrent risk for HIV, one might then transition. Now, remember that PEP involves 3 medications because we don't have preventive medicine onboard before the person is exposed, and we think that it's helpful to take 3 medicines because of the large inoculum without any prior protection. Animal data suggests a 28-day regimen. Then if the individual is going to continue to be at-risk for HIV after the initial PEP dosage, one may want to transition to the fixed-dose combination of TDF/FTC for pre-exposure prophylaxis for the future.


|
|
| |
| |
|
|
|