| |
|
Acute HCV & PrEP/MSM HIV+ & HIV-Neg
|
| |
| |
Download the PDF here
Patterns of Hepatitis C Virus Transmission in Human Immunodeficiency Virus (HIV)-infected and HIV-negative Men Who Have Sex With Men
From 2014 to 2017, 108 AHIs (80 first infections, 28 reinfections) were reported in 96 MSM (HIV-infected, 72; HIV-negative, 24).
AHI incidence rose from 1.1/100 person-years (95 confidence interval [CI], 0.7-1.7) in 2014 to 2.4/100 person-years (95 CI, 1.1-2.6) in 2017 in HIV-infected MSM (P = .05) and from 0.3/100 person-years (95 CI, 0.06-1.0) in 2016 to 3.4/100 person-years (95 CI, 2.0-5.5) in 2017 in PrEP users (P < .001). Eleven clusters were identified. All clusters included HIV-infected MSM; 6 also included HIV-negative MSM. All clusters started with ≥1 HIV-infected MSM. Risk factor distribution varied among clusters.
Yesterday I reported increased Chemsex in the HIV Swiss Cohort, published data, new. Suggesting we may see increased acute HCV infections. I predicted this 7 years go but no one wanted to listen including local govt officials.
High incidence of HCV (re-)infections among PrEP users in the Netherlands: implications for prevention, monitoring and treatment
HCV incidence in HIV-infected and in PrEP-using MSM - New HCV Rate Similar in HIV+ MSM and HIV- MSM on PrEP in France
EACS/2019: High HCV re infection rate in MSM living with HIV in Barcelona: The need to focus on high risk population to achieve HCV elimination ......Conclusions: High rate of HCV reinfection has been observed in MSM LWHIV in our cohort, most of them being asymptomatic and mainly associated to other concomitant STI.
HCV-RNA should be tested regularly in HIV-positive MSM already cured from an AHC and routine screening of HCV should be done when an STI is diagnosed. Assessment of high risk factors must be done, and risk reduction strategies are needed in this population .
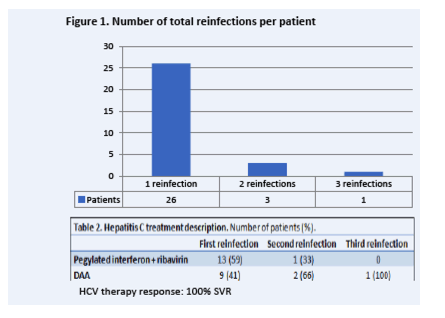
"higher incidence in both HIV-infected and in PrEP-using MSM in this study than recently reported at the national level in the French Dat’AIDS cohort.....Based on these observations, it appears essential to offer systematic and frequent HCV screening among MSM reporting high-risk practices regardless of HIV status.....All clusters started with an HIV-infected MSM, suggesting that the epidemic started in this population and later spread to HIV-negative MSM.....Our cohort study clearly demonstrates that the HCV epidemic has now spread in HIV-negative MSM, whereas this population had been relatively spared until recently [2, 14, 15]. The increase of AHI incidence was particularly high in PrEP-using MSM, which may be related either to an increase in high-risk practices in these patients, who represented 67% of AHI cases among HIV-negative MSM, or to better identification of cases through systematic screening. Indeed, HCV screening is performed every 3 months in PrEP-using MSM, whereas HIV-negative MSM not using PrEP are only tested when spontaneously attending STI clinics or in case of symptomatic hepatitis. Therefore, diagnosis in HIV-negative MSM not using PrEP could be delayed and could fuel the epidemic in other groups of patients....Previously reported risk factors were identified in this cohort (IV and nasal drug use, sex parties, traumatic sexual practices) [16]. At least 1 factor was reported in 79% and 96% of AHI cases in HIV-infected and HIV-negative MSM, respectively. Specific risk factors were overrepresented in some clusters, notably fisting and IV drug use in cluster C1 and nasal drug use in cluster C3, and are presumed to be responsible for the large number of AHIs observed in these clusters in 2016 and 2017.......As previously reported, treatment response with DAA during the acute phase was similar to the response during chronic infection [23-25]. However, efforts are needed to shorten the delay from diagnosis to treatment, since the median time from first detectable HCV-RNA to treatment was longer than 5 months. Increasing the screening frequency could shorten this delay in the most at-risk patients, as recommended in PrEP users. However, the 3-month silent serological window can further differ diagnosis unless HCV-RNA is used for screening, which is not routinely recommended except in patients cured from a previous hepatitis.....Based on these observations, it appears essential to offer systematic and frequent HCV screening among MSM reporting high-risk practices regardless of HIV status. DAA treatment and harm reduction interventions should be offered early following the diagnosis of AHI in this population in order to limit the epidemic spread and avoid reinfections"
Patterns of Hepatitis C Virus Transmission in Human Immunodeficiency Virus (HIV)-infected and HIV-negative Men Who Have Sex With Men
Christophe Ramiere,1,2,3,a Caroline Charre,1,3,8,a Patrick Miailhes,4 François Bailly,5 Sylvie Radenne,5 Anne-Claire Uhres,6 Corinne Brochier,7
Matthieu Godinot,4 Pierre Chiarello,4 Pierre Pradat,7 and Laurent Cotte4,8; for the Lyon Acute Hepatitis Study Groupb
1Virology Laboratory, Hospices Civils de Lyon, Hôpital de la Croix-Rousse, 2Centre International de Recherche en Infectiologie, INSERM U1111, CNRS UMR 5308, 3University of Lyon, Universite Claude Bernard Lyon1, Villeurbanne, 4Infectious Diseases Department, 5Hepatology Department, 6Pharmacy, and 7Clinical Research Centre, Hospices Civils de Lyon, Hôpital de la Croix-Rousse, and 8INSERM U1052, Lyon, France - Clinical Infectious Diseases 27 February 2019
The introduction of preexposure prophylaxis (PrEP) as a prevention tool for HIV raised concerns about the risk of sexually transmitted infections (STIs), including HCV, in patients engaged in high-risk activities [3]. We recently reported that the incidence of acute HCV infection (AHI) in 2016-2017 in France was similar in HIV-infected MSM (1.2/100 person-years) and in HIV-negative MSM receiving PrEP (1.2/100 person-years) [4]. Evidence of HCV transmission from HIV-infected to HIV-negative MSM was also recently reported [5, 6], suggesting that the HCV epidemic was spreading from the former population to the latter.
The number of AHI cases sharply increased in Lyon between 2014 and 2017 both in HIV-infected and in HIV-negative MSM. This increase was mainly related to an increase in first infections in HIV-negative MSM. First infections remained stable in HIV-infected MSM, but reinfections sharply increased in this population, suggesting that the number of patients engaged in high-risk activities was stable, while a significant number of patients pursued high-risk activities following a first infection. This resulted in a higher incidence in both HIV-infected and in PrEP-using MSM in this study than recently reported at the national level in the French Dat’AIDS cohort [4].
Abstract
Background
Sexually transmitted acute hepatitis C virus (HCV) infections (AHIs) have been mainly described in human immunodeficiency virus (HIV)-infected men who have sex with men (MSM). Cases in HIV-negative MSM are scarce. We describe the epidemic of AHI in HIV-infected and HIV-negative MSM in Lyon, France.
Methods
All cases of AHI diagnosed in MSM in Lyon University Hospital from 2014 to 2017 were included. AHI incidence was determined in HIV-infected and in preexposure prophylaxis (PrEP)-using MSM. Transmission clusters were identified by construction of phylogenetic trees based on HCV NS5B (genotype 1a/4d) or NS5A (genotype 3a) Sanger sequencing.
Results
From 2014 to 2017, 108 AHIs (80 first infections, 28 reinfections) were reported in 96 MSM (HIV-infected, 72; HIV-negative, 24).
AHI incidence rose from 1.1/100 person-years (95 confidence interval [CI], 0.7-1.7) in 2014 to 2.4/100 person-years (95 CI, 1.1-2.6) in 2017 in HIV-infected MSM (P = .05) and from 0.3/100 person-years (95 CI, 0.06-1.0) in 2016 to 3.4/100 person-years (95 CI, 2.0-5.5) in 2017 in PrEP users (P < .001). Eleven clusters were identified. All clusters included HIV-infected MSM; 6 also included HIV-negative MSM. All clusters started with ≥1 HIV-infected MSM. Risk factor distribution varied among clusters.
Conclusions
AHI incidence increased in both HIV-infected and HIV-negative MSM. Cluster analysis suggests initial transmission from HIV-infected to HIV-negative MSM through chemsex and traumatic sexual practices, leading to mixed patterns of transmission regardless of HIV status and no overlap with the general population.
| |
| |
| |
|
|
|