| |
|
Clinical outcomes in patients with chronic hepatitis C after direct-acting antiviral treatment: a prospective cohort study - decrease mortality/ HCC
|
| |
| |
Download the PDF
DAAs/SVR Large French ANRS Cohort - "decrease in all-cause mortality (HR 0⋅48, 95% CI 0⋅33-0⋅70; p=0⋅0001), liver-related death (0⋅39, 0⋅21-0⋅71; p=0⋅0020), non-liver-related death (0⋅60, 0⋅36-1⋅00; p=0⋅048), and hepatocellular carcinoma (0⋅66, 0⋅46-0⋅93; p=0⋅018), and was no longer associated with decompensated cirrhosis (1⋅14, 0⋅57-2⋅27; p=0⋅72)."
"The findings of this large French cohort study show that direct-acting antiviral treatment is associated with reduced risk for mortality and hepatocellular carcinoma, after adjustment for potential confounding factors. Similar associations were identified in the subgroup of patients with cirrhosis. These inverse associations persisted in the subgroup of patients who achieved a sustained virological response, whereas those who did not achieve a sustained virological response were at higher risk for hepatocellular carcinoma. No signs were seen of increased risk for hepatocellular carcinoma during direct-acting antiviral treatment.....A striking finding in our study was the lower risk for non-liver-related mortality in patients treated with direct-acting antivirals compared with untreated patients."
"After adjustment in the multivariable analysis, exposure to direct-acting antivirals was associated with a decrease in all-cause mortality (HR 0⋅48, 95% CI 0⋅33-0⋅70; p=0⋅0001), liver-related death (0⋅39, 0⋅21-0⋅71; p=0⋅0020), non-liver-related death (0⋅60, 0⋅36-1⋅00; p=0⋅048), and hepatocellular carcinoma (0⋅66, 0⋅46-0⋅93; p=0⋅018), and was no longer associated with decompensated cirrhosis (1⋅14, 0⋅57-2⋅27; p=0⋅72)."
"In adjusted multivariable analyses in 3045 patients with baseline cirrhosis, exposure to direct-acting antivirals was strongly associated with a decrease in all-cause mortality (HR 0⋅34, 95% CI 0⋅22-0⋅55; p<0⋅0001), liver-related mortality (0⋅28, 0⋅15-0⋅54; p=0⋅0001), non-liver-related mortality (0⋅40, 0⋅19-0⋅83; p=0⋅015), and hepatocellular carcinoma (0⋅57, 0⋅40-0⋅81; p=0⋅0016; table 2; figure 3)."
Clinical outcomes in patients with chronic hepatitis C after direct-acting antiviral treatment: a prospective cohort study
Lancet Feb 11 2019
Fabrice Carrat, Helene Fontaine, Celine Dorival, Melanie Simony, Alpha Diallo, Christophe Hezode, Victor De Ledinghen, Dominique Larrey, Georges Haour, Jean-Pierre Bronowicki, Fabien Zoulim, Tarik Asselah, Patrick Marcellin, Dominique Thabut, Vincent Leroy, Albert Tran, François Habersetzer, Didier Samuel, Dominique Guyader, Olivier Chazouilleres, Philippe Mathurin, Sophie Metivier, Laurent Alric, Ghassan Riachi, Jerôme Gournay, Armand Abergel, Paul Cales, Nathalie Ganne, Veronique Loustaud-Ratti, Louis D'Alteroche, Xavier Causse, Claire Geist, Anne Minello, Isabelle Rosa, Moana Gelu-Simeon, Isabelle Portal, François Raffi, Marc Bourliere, Stanislas Pol, for the French ANRS CO22 Hepather cohort*
Summary
Background
Although direct-acting antivirals have been used extensively to treat patients with chronic hepatitis C virus (HCV) infection, their clinical effectiveness has not been well reported.
We compared the incidence of death, hepatocellular carcinoma, and decompensated cirrhosis between patients treated with direct-acting antivirals and those untreated, in the French ANRS CO22 Hepather cohort.
Methods
We did a prospective study in adult patients with chronic HCV infection enrolled from 32 expert hepatology centres in France.
We excluded patients with chronic hepatitis B, those with a history of decompensated cirrhosis, hepatocellular carcinoma, or liver transplantation, and patients who were treated with interferon-ribavirin with or without first-generation protease inhibitors. Co-primary study outcomes were incidence of all-cause mortality, hepatocellular carcinoma, and decompensated cirrhosis. The association between direct-acting antivirals and these outcomes was quantified using time-dependent Cox proportional hazards models. This study is registered with ClinicalTrials.gov, number NCT01953458.
Findings
Between Aug 6, 2012, and Dec 31, 2015, 10 166 patients were eligible for the study. 9895 (97%) patients had available follow-up information and were included in analyses.
Median follow-up was 33⋅4 months (IQR 24⋅0-40⋅7). Treatment with direct-acting antivirals was initiated during follow-up in 7344 patients, and 2551 patients remained untreated at the final follow-up visit.
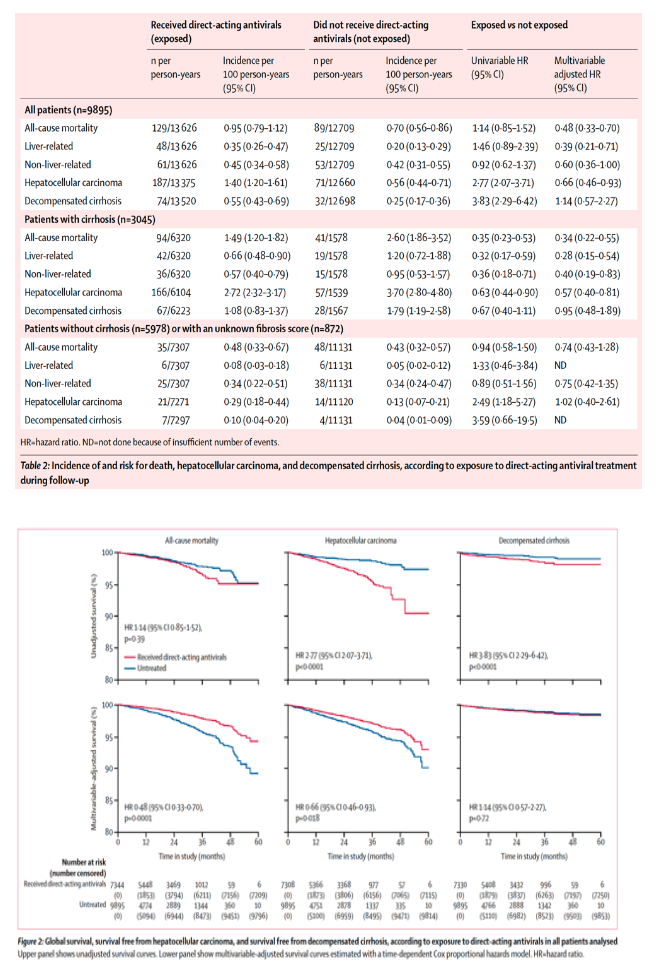
During follow-up, 218 patients died (129 treated, 89 untreated), 258 reported hepatocellular carcinoma (187 treated, 71 untreated), and 106 had decompensated cirrhosis (74 treated, 32 untreated).
Exposure to direct-acting antivirals was associated with increased risk for hepatocellular carcinoma (unadjusted hazard ratio [HR] 2⋅77, 95% CI 2⋅07-3⋅71) and decompensated cirrhosis (3⋅83, 2⋅29-6⋅42).
After adjustment for variables (age, sex, body-mass index, geographical origin, infection route, fibrosis score, HCV treatment-naive, HCV genotype, alcohol consumption, diabetes, arterial hypertension, biological variables, and model for end-stage liver disease score in patients with cirrhosis), exposure to direct-acting antivirals was associated with a decrease in all-cause mortality (adjusted HR 0⋅48, 95% CI 0⋅33-0⋅70) and hepatocellular carcinoma (0⋅66, 0⋅46-0⋅93), and was not associated with decompensated cirrhosis (1⋅14, 0⋅57-2⋅27).
Interpretation
Treatment with direct-acting antivirals is associated with reduced risk for mortality and hepatocellular carcinoma and should be considered in all patients with chronic HCV infection.
Funding
INSERM-ANRS (France Recherche Nord & Sud Sida-HIV Hepatites), ANR (Agence Nationale de la Recherche), DGS (Direction Generale de la Sante), MSD, Janssen, Gilead, AbbVie, Bristol-Myers Squibb, and Roche.
Findings of observational studies in patients with HCV infection show reduced risk for hepatocellular carcinoma, complications of liver disease, and mortality in patients treated with interferon or direct-acting antivirals who achieve a sustained virological response.
However, very few studies have compared the clinical outcomes of patients treated and not treated with direct-acting antivirals, as would be done in a randomised trial.
The findings of a single-centre cohort study showed decreased mortality in patients receiving a combination of paritaprevir, ritonavir, ombitasvir, and dasabuvir, or of sofosbuvir and ledispasvir, compared with untreated patients.
However, this study did not report the incidence of liver-related events such as liver decompensation or hepatocellular carcinoma, and this information is important because of controversy surrounding a potential increase in risk for hepatocellular carcinoma with direct-acting antiviral treatment.
The aim of this study was to further clarify the benefits or harms of direct-acting antivirals by comparing the incidence of death, hepatocellular carcinoma, and decompensated cirrhosis in patients treated with direct-acting antivirals and untreated, from the prospective French ANRS CO22 Hepather cohort.
Study design and participants
The ANRS CO22 Hepather cohort is a French national, multicentre, prospective, observational cohort study of patients with active or inactive hepatitis B virus (HBV) or past or present HCV infection, which started in August, 2012.
The main objectives of the study are to quantify the clinical efficacy and safety of new hepatitis treatments in real life. The anticipated cohort size is 15 000 patients serum-positive for anti-HCV (>90% with existing chronic HCV infection at entry—ie, serum-positive for HCV-RNA—and <10% with past chronic HCV infection) and 10 000 patients with active or inactive chronic HBV infection, to be followed up for a median of 7 years. Main exclusion criteria are HIV co-infection and ongoing treatment for HCV infection at inclusion. In the current analysis, we selected all patients with chronic HCV infection at entry. Participants were recruited consecutively during a medical visit at one of 32 expert hepatology centres in France.
Results
Between Aug 6, 2012, and Dec 31, 2015, 14 389 anti-HCV-positive patients had been recruited to the ANRS CO22 Hepather cohort, including 11 870 with chronic HCV infection at entry (figure 1). 95 patients had active HBV co-infection at entry, 653 had a history of hepatocellular carcinoma, 1003 had decompensated cirrhosis, and 326 had undergone liver transplantation; these patients were excluded from this study. A further 148 patients were excluded who had received peginterferon and ribavirin with or without a first-generation protease inhibitor after entry in the cohort.
10 166 patients were judged eligible for this study (figure 1). Follow-up information was missing for 271 patients, therefore post-entry follow-up information was available for 9895 (97%) patients, who were included in analyses. The appendix presents a comparison of characteristics of eligible patients with missing follow-up information.
Of 9895 patients analysed, 1326 (13%) had a platelet count lower than 150 000 platelets per μL or a prothrombin time less than 70% and were considered to have cirrhosis. In other patients, fibrosis was assessed by liver biopsy for 398 (4%) patients, by Fibroscan for 3188 (32%), by Fibrotest for 1812 (18%), by Fibrometer for 635 (6%), and by the Hepascore for 143 (1%). The clinician assessed the level of fibrosis for 1521 (15%) patients, and the baseline fibrosis score remained unknown in 872 (9%). In total, 3045 (31%) patients had cirrhosis. The median time between assessment of fibrosis and end of follow-up in patients who received direct-acting antivirals was 34⋅5 months (IQR 25⋅1-43⋅0), and for those who were untreated, median time was 32⋅3 months (IQR 22⋅7-43⋅0).
Baseline demographic, clinical, and laboratory characteristics of included patients, according to exposure to direct-acting antivirals during follow-up, are provided in table 1. Median patients' age was 56⋅0 years (IQR 50⋅0-64⋅0) and 5279 (53%) patients were men. 7344 patients began direct-acting antiviral treatment after a median time from entry of 4⋅3 months (IQR 0⋅2-17⋅2). Median follow-up (untreated plus treated periods) in these patients was 33⋅4 months (IQR 24⋅0-40⋅7). At the last follow-up visit, 2551 patients remained untreated, with a median follow-up of 31⋅2 months (IQR 21⋅5-41⋅0). Patients who received direct-acting antivirals were older, more frequently men, had a higher body-mass index, and reported past excessive alcohol use compared with those who remained untreated at the final follow-up visit. Receiving direct-acting antiviral treatment was also strongly associated with the severity of liver disease and other comorbidities (table 1).
Compared with untreated patients, those who received direct-acting antivirals had been diagnosed with HCV for a longer time, 2823 (42%) of 6800 patients had cirrhosis (vs 222 [10%] of 2223 untreated patients), 4159 (57%) of 7324 patients had received HCV treatment at entry (vs 974 [39%] of 2516 untreated patients), including 49 (1%) of 7324 with past use of interferon-free regimens (vs three [<1%] of 2516 untreated patients), 918 (13%) of 7227 patients were infected with HCV genotype 3 (vs 211 [9%] of 2375 untreated patients), 945 (13%) of 7270 patients had diabetes (vs 198 [8%] of 2475 untreated patients), and 2161 (30%) of 7266 patients had arterial hypertension (vs 589 [24%] of 2467 untreated patients). Of note, 1049 (40%) of 2607 patients with past excessive alcohol use had cirrhosis versus 1977 (28%) of 7119 without (p<0⋅0001), which could be why past excessive alcohol users were more likely to initiate direct-acting antiviral treatment. Combinations of direct-acting antivirals used in the study are listed in the appendix.
218 patients died during the study; 73 were classified as liver-related deaths, 114 as non-liver-related (appendix), and 31 deaths were unclassified. 258 cases of hepatocellular carcinoma and 106 cases of decompensated cirrhosis were reported during follow-up (figure 1). 25 patients also underwent liver transplantation during follow-up. The crude incidence of all-cause mortality, liver-related death, hepatocellular carcinoma, and decompensated cirrhosis was higher in patients exposed to direct-acting antivirals than in unexposed patients (table 2; figure 2). Exposure to direct-acting antivirals was associated with increased risk for hepatocellular carcinoma (HR 2⋅77, 95% CI 2⋅07-3⋅71; p<0⋅0001) and decompensated cirrhosis (3⋅83, 2⋅29-6⋅42; p<0⋅0001) in the unadjusted Cox model.
After adjustment in the multivariable analysis, exposure to direct-acting antivirals was associated with a decrease in all-cause mortality (HR 0⋅48, 95% CI 0⋅33-0⋅70; p=0⋅0001), liver-related death (0⋅39, 0⋅21-0⋅71; p=0⋅0020), non-liver-related death (0⋅60, 0⋅36-1⋅00; p=0⋅048), and hepatocellular carcinoma (0⋅66, 0⋅46-0⋅93; p=0⋅018), and was no longer associated with decompensated cirrhosis (1⋅14, 0⋅57-2⋅27; p=0⋅72).
Similar findings were obtained using inverse probability of treatment weighting and weighted sequential Cox models or when analyses were restricted to events that occurred after 12 months of follow-up (appendix). Other predictors independently associated with risk for all-cause mortality, hepatocellular carcinoma, or decompensated cirrhosis are presented in table 3 and include cirrhosis, markers of liver failure, hypertension, anaemia, and serum α-fetoprotein; a description of events by covariate levels is provided in the appendix.
A sustained virological response was achieved by 5615 (76%) of 7344 patients who started direct-acting antivirals, was not achieved by 341 (5%) patients, and was unknown in 709 (10%) patients; sustained virological response status could not be established for 679 (9%) patients because of insufficient follow-up (these patients are classified as on-treatment; appendix). Thus, the proportion of patients achieving a sustained virological response was 94% (5615 of 5956 patients who had known status and sufficient follow-up). In the adjusted multivariable analysis, compared with untreated patients, achieving a sustained virological response in patients who received direct-acting antivirals was associated with a decrease in all-cause mortality, liver-related mortality, non-liver-related mortality, and hepatocellular carcinoma, and with a non-significant decrease in decompensated cirrhosis, whereas not achieving a sustained virological response was associated with a significant increase in hepatocellular carcinoma (adjusted HR 2⋅23, 95% CI 1⋅37-3⋅64; p=0⋅0012; appendix). The median time between assessment of a sustained virological response and diagnosis of hepatocellular carcinoma was 14⋅0 months (IQR 7⋅4-21⋅1) in patients without a sustained virological response and 12⋅1 months (5⋅9-20⋅1) in those with a sustained virological response (p=0⋅29). No evidence was found of increased risk for hepatocellular carcinoma during the on-treatment period (adjusted HR 0⋅74, 95% CI 0⋅49-1⋅13; p=0⋅17).
In adjusted multivariable analyses in 3045 patients with baseline cirrhosis, exposure to direct-acting antivirals was strongly associated with a decrease in all-cause mortality (HR 0⋅34, 95% CI 0⋅22-0⋅55; p<0⋅0001), liver-related mortality (0⋅28, 0⋅15-0⋅54; p=0⋅0001), non-liver-related mortality (0⋅40, 0⋅19-0⋅83; p=0⋅015), and hepatocellular carcinoma (0⋅57, 0⋅40-0⋅81; p=0⋅0016; table 2; figure 3). Predictors of clinical events in patients with cirrhosis were similar to those identified in the entire cohort (appendix). A sustained virological response was achieved by 2329 (83%) of 2823 patients with cirrhosis who initiated direct-acting antivirals, was not achieved by 195 (7%) patients, and was unknown in 179 (6%) patients; 120 (4%) patients were still on-treatment. Thus, the proportion of patients with cirrhosis achieving a sustained virological response was 92% (2329 of 2524 patients who had known status and sufficient follow-up). Multivariable analyses confirmed the association between achieving a sustained virological response and a decrease in all-cause mortality, liver-related mortality, non-liver-related mortality, and hepatocellular carcinoma. The association was also confirmed between not achieving a sustained virological response and increased risk for hepatocellular carcinoma (appendix).
We did not find any association between exposure to direct-acting antivirals and mortality and clinical outcomes in the subset of patients without cirrhosis or with an unknown fibrosis score at entry (table 2). A sustained virological response was achieved by 3286 (73%) of 4521 patients who initiated direct-acting antivirals, was not achieved by 146 (3%), and was unknown in 530 (12%); 559 (12%) patients were still on-treatment. Thus, the proportion of patients without cirrhosis or with an unknown fibrosis score who achieved a sustained virological response was 96% (3286 of 3432 patients who had known status and sufficient follow-up).
Detailed characteristics of hepatocellular carcinoma were obtained for 249 (97%) of 258 patients with incident hepatocellular carcinoma. No difference was found between patients treated with direct-acting antivirals and untreated patients in the delay between the last normal imaging test and diagnosis, macroscopic pattern, number of tumours at diagnosis, total nodule size, largest nodule size, or serum α-fetoprotein (appendix).
Discussion
The findings of this large French cohort study show that direct-acting antiviral treatment is associated with reduced risk for mortality and hepatocellular carcinoma, after adjustment for potential confounding factors. Similar associations were identified in the subgroup of patients with cirrhosis. These inverse associations persisted in the subgroup of patients who achieved a sustained virological response, whereas those who did not achieve a sustained virological response were at higher risk for hepatocellular carcinoma. No signs were seen of increased risk for hepatocellular carcinoma during direct-acting antiviral treatment.
Overall, our results are similar to those reported in the ERCHIVES retrospective cohort.
In that study, a significant (57%, 95% CI 43-67) decrease in all-cause mortality was noted in patients receiving direct-acting antivirals compared with propensity-score-matched untreated patients. Moreover, results showed that age, cirrhosis, comorbidities (diabetes and chronic kidney disease), and anaemia were positively correlated with mortality. In our study, we reported a strong independent relation between all-cause mortality and cirrhosis, markers of liver failure, hypertension, and anaemia. Our results also confirm those of studies in which a lower risk of death was noted in patients treated with direct-acting antivirals who achieved a sustained virological response, compared with those who did not achieve a sustained virological response.
The incidence of hepatocellular carcinoma in patients with a sustained virological response after treatment with direct-acting antivirals in our study (appendix) overlapped with the incidence reported in another study (1⋅15 [95% CI 0⋅93-1⋅41] per 100 person-years vs 0⋅90 [0⋅77-1⋅03] per 100 person-years),
whereas incidence in patients without a sustained virological response was significantly higher in our study compared with this other study (7⋅19 [95% CI 5⋅16-9⋅76] per 100 person-years vs 3⋅45 [2⋅73-4⋅18] per 100 person-years). This difference could be because patients at highest risk for hepatic morbidity and mortality (eg, those with a history of excessive alcohol use) received direct-acting antivirals in our study.
A striking finding in our study was the lower risk for non-liver-related mortality in patients treated with direct-acting antivirals compared with untreated patients. Although a decrease in long-term non-liver-related mortality has been reported in patients with sustained virological response compared with those without a sustained virological response after interferon-based therapy,
reverse causality could be another possibility if patients with the most severe liver disease and the highest risk for death from any cause had a lower probability of starting direct-acting antiviral treatment. However, patients with decompensated cirrhosis or a history of hepatocellular carcinoma were excluded at baseline. We adjusted for many markers of liver insufficiency and comorbidities in our multivariable analyses. Finally, our results were similar when data from the first 12 months of follow-up were excluded. These elements seem to exclude reverse causality.
Our study has several limitations. First, the assessment of fibrosis and cirrhosis was based on patients' records at entry in the cohort, ascertained by different methods, and not updated during follow-up or when patients started direct-acting antiviral treatment. We validated the predictive value of platelet count and prothrombin time for diagnosis of cirrhosis, using other methods for the assessment of fibrosis. The median time between assessment of fibrosis and end of follow-up did not differ between untreated and treated patients. Fibrosis probably worsened in some patients, thus accounting for the development of liver-related complications in patients classified as without cirrhosis at entry in the cohort. However, any difference in the progression of fibrosis between patients untreated and treated with direct-acting antivirals during follow-up would be directly attributable to the effect of treatment on fibrosis. Thus, lack of assessment of fibrosis during follow-up should not be regarded as a bias but rather a plausible explanation for the inverse relation between treatment and risk for liver-related outcomes. Moreover, results in the subgroup of patients with baseline cirrhosis, which should be less biased by the misclassification of fibrosis, were consistent.
A second limitation of our study is that the duration of follow-up was short, making assessment of long-term outcomes associated with direct-acting antivirals impossible. Nevertheless, an inverse relation was noted between treatment with direct-acting antivirals and liver-related mortality or hepatocellular carcinoma in patients with cirrhosis over this short-term follow-up period, and a longer duration of follow-up would probably not change these findings.
Third, because of the observational nature of our study, some patients might have undergone less regular screening for hepatocellular carcinoma than recommended, resulting in potentially missed diagnoses. However, the average number of follow-up visits and ultrasound examinations (weighted by person-years of follow-up) were higher in patients during treatment and the year after treatment than in untreated patients or before treatment (data available on request). Therefore, any screening bias would result in a decrease in detection of hepatocellular carcinoma in patients not treated with direct-acting antivirals, compared with treated patients, and would not affect our conclusions.
A fourth limitation is that no association was seen between direct-acting antivirals and risk for decompensated cirrhosis. However, the analysis according to sustained virological response status (appendix) shows a non-significant inverse association in patients with cirrhosis at baseline (patients treated with direct-acting antivirals who achieved a sustained virological response vs untreated patients, HR 0⋅51, 95% CI 0⋅23-1⋅14), and our study probably does not have statistical power for this outcome.
Finally, although many multivariable analyses were done, we cannot exclude either a residual risk of bias from confounding factors associated with unmeasured prognostic factors or another complex time-dependent selection bias. We used different statistical methods to account for these different sources of bias, with similar results.
Because of the observational design of our study, we cannot formally conclude that inverse associations between direct-acting antiviral treatment and mortality or incidence of hepatocellular carcinoma reflect cause and effect relations. However, we can postulate about plausible mechanisms. Direct-acting antivirals induce a sustained virological response, reducing liver damage and inflammation. This effect causes liver regeneration, decreasing risk for progression to liver-related complications or hepatocellular carcinoma. Our results showing strikingly different risks for these liver-related events in patients with and without a sustained virological response support these mechanisms. Researchers have also suggested that not achieving a sustained virological response could be a sign of hepatocellular carcinoma.
However, the median time between assessment of sustained virological response and diagnosis of hepatocellular carcinoma did not differ between patients with and without a sustained virological response. This finding does not support the presence of pre-existing hepatocellular carcinoma in patients without a sustained virological response.
In summary, the findings of this large prospective cohort study showed a significant decrease in risk for all-cause mortality and hepatocellular carcinoma associated with direct-acting antiviral treatment. Our results also suggest that direct-acting antivirals do not adversely affect the development of hepatocellular carcinoma. The long-term effects of direct-acting antivirals on liver decompensation must still be clarified.
| |
| |
| |
|
|
|