| |
Congestive Heart Failure and Chronic Kidney Disease
The CardioRenal/NephroCardiology Connection - EDITORIAL COMMENT
|
| |
| |
Download the PDF here
Charles A. Herzog
Cardiac disease and chronic kidney disease (CKD) are inextricably linked. The graded risk of cardiovascular morbidity and mortality is strongly related to both declining estimated glomerular filtration rate (eGFR) and severity of albuminuria. This is evident for patients with CKD and heart failure; incidence of heart failure was 3-fold higher in subjects in the ARIC (Atherosclerosis Risk In Communities) study who had eGFRs of <60 ml/min/1.73 m2 than for those with eGFRs of ≥90 ml/min/1.73 m2 (1). Reduced eGFR and albuminuria are associated with increased mortality and heart failure hospitalizations in individuals with heart failure with reduced or preserved left ventricular (LV) systolic function (2,3). Moreover, the triad of CKD, heart failure, and diabetes may coexist in older adult patients, and each condition is responsible for a disproportionately large percentage of Medicare expenditures (Figure 1).
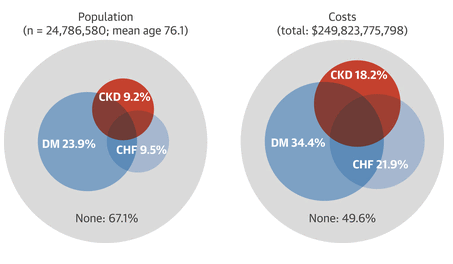
Figure 1
Relative Proportions of Medicare Beneficiaries and Percentages of Medicare Expenditures for Diabetes, CHF, and CKD
Point prevalent distribution and annual costs of Medicare (fee-for-service) patients, age 65 years or older, with diagnosed diabetes mellitus (DM), congestive heart failure (CHF), and chronic kidney disease (CKD), 2011. Source: United States Renal Data System. 2013 Annual Data Report: Epidemiology of Kidney Disease in the United States. Bethesda, MD: National Institutes of Health, National Institute of Diabetes and Digestive and Kidney Diseases, 2013: volume 1, Figure 7.1.
Understanding of the linked epidemiology of CKD, heart failure, (and diabetes) has been fostered by elegant long-term prospective cohort studies, including ARIC, and especially the CRIC (Chronic Renal Insufficiency Cohort) study. The CRIC cohort continues to inform about the intricate bidirectional interplay of CKD and heart disease over time and provides invaluable data for planning clinical trials designed to help patients with CKD and heart disease (including heart failure). Large observational data sets derived from administrative data (e.g., the U.S. Renal Data System) provide a real-world complement to amplify understanding of the cardiorenal connection.
The paper by Bansal et al. (4) in this issue of the Journal is a distinguished contribution to the literature based on CRIC data; this study enhances understanding of the intertwined CKD and heart failure hospitalization risk. The investigators provided data on heart failure hospitalizations (in reduced and preserved LV systolic performance) related to eGFR and severity of albuminuria, and also provided new data on heart failure readmission rates. From a cardiorenal perspective, the most interesting data pertain to the 2-fold increased adjusted hazard for CKD progression (and all-cause death) in participants hospitalized for heart failure within 2 years of study entry (and 3-fold increased mortality risk in those with ≥2 heart failure admissions). For the entire cohort, the crude heart failure admission rate was 5.8 per 100 patient-years (higher for preserved LV ejection fraction [LVEF] vs. reduced LVEF, which reflected higher differential rates for first, but not subsequent, admissions in patients with preserved LVEF). The graded increased adjusted heart failure admission rates were related to reduced eGFRs (rate ratio: 1.7; 95% confidence interval [CI]: 1.3 to 2.2) for 30 to 44 ml/min/1.73 m2 and a rate ratio of 2.2 (95% CI: 1.7 to 2.9) for <30 ml/min/1.73 m2 versus >45 ml/min/1.73 m2. Higher urine albumin-to-creatine ratios (ACRs) (30 to 299 and ≥300 mg/kg) were respectively associated with rate ratios of 1.9 (95% CI: 1.5 to 2.6) and 2.6 (95% CI: 1.9 to 3.5) compared with an ACR of <30 mg/kg. In the entire study cohort, 20.6% were subsequently readmitted for heart failure within 30 days. Finally, the number of heart failure hospitalization days (which occurred in the first 2 study years) was strongly correlated with CKD progression (eGFR decline of at least 50% or progression to end-stage kidney disease [ESKD]) and death. Adjusted hazard ratios were 1.90 (95% CI: 1.15 to 3.15) for CKD progression and 2.91 (95% CI: 2.17 to 3.90) for death for participants with ≥8 heart failure hospitalization days (vs. none).
The final analytical cohort for the Bansal et al. study included 3,791 participants, 48% of whom had diabetes. Across the cohort, 43% of participants with no heart failure events had diabetes, as did 71% of those with >0 to 1 heart failure event per year, and 70% of those with >1 event per year. This was not surprising because of the high risk of heart failure associated with diabetes, but clinically, it is exceedingly important in the context of sodium-glucose cotransporter 2 inhibitor (SGLT2i) therapy used to reduce the risk of incident and prevalent heart failure and CKD progression in patients with type 2 diabetes (5). In the Bansal et al. study, 45% of participants had eGFRs of ≥45 ml/min/1.73 m2 (the eGFR breakdown was unknown for diabetic participants). Plausibly, some (perhaps, one-third?) of this group might be candidates for SGLT2i agents under current treatment guidelines. Moreover, an additional 1,379 participants had eGFRs of 30 to 44 ml/min/1.73 m2 (the number of participants with type 2 diabetes can be guessed) and did not qualify for SGLT2i treatment. That might change after publication of the CREDENCE (Canagliflozin and Renal Events in Diabetes with Established Nephropathy Clinical Evaluation; NCT02065791) trial, which enrolled patients with diabetes and established kidney disease (eGFR ≥30 to <90 ml/min/1.73 m2). This trial, which was published on April 14, 2019, found a 30% (hazard ratio: 0.70; 95% CI: 0.59 to 0.82) lower relative risk for canagliflozin versus placebo for the primary outcome of a composite of ESKD (time to dialysis or kidney transplantation), doubling of serum creatinine, and renal or cardiovascular death, when used in addition to standard care (6). The EMPA-KIDNEY (Study of Heart and Kidney Protection with Empagliflozin; NCT03594110), starting in 2019, will test the efficacy of empagliflozin (vs. placebo) for prevention of the primary composite outcome of CKD progression or cardiovascular death in CKD patients with eGFRs of ≥20 to <45 ml/min/1.73 m2 or ≥45 to <90 ml/min/1.73 m2 and albuminuria (ACR ≥200 mg/g) (i.e., not dissimilar to the cohort in the Bansal et al. [4] analytical cohort).
If you have not yet read the 2018 American College of Cardiology Expert Consensus Decision Pathway on Novel Therapies for Cardiovascular Risk Reduction in Patients With Type 2 Diabetes and Atherosclerotic Cardiovascular Disease (7), please set aside this editorial now, and carefully read this remarkable, informative document, then return after pondering this exhortation: "Given the data supporting comprehensive CV risk reduction in patients with T2D, CV clinicians should be both champions and change agents as strong advocates for our patients, recognizing unmet needs in health care delivery, and extending our comfort zone in implementing the use of new evidence-based therapies that reduce CV event rates" (7). The document addresses implementation issues ("systems factors"). I would endorse the comprehensive team approach (including nephrologists, who somehow were not included). Approximately one-half of the Bansal et al. (4) analytical cohort would qualify for this team approach (to type 2 diabetes), but, from a nephrocardiology perspective, the nondiabetic one-half also needs a team approach based on their formidable risk for cardiovascular disease.
Methodologically, one strength of the Bansal et al. (4) study is that heart failure events were formally adjudicated (and confirmed "based on modified clinical Framingham criteria") in a study cohort of participants with mild to moderate CKD. However, heart failure adjudication in patients with heart disease with stage 5 CKD who receive conventional thrice weekly hemodialysis may be a methodologically more challenging Herculean task than cleaning the Augean stables in 1 day. Phenotypically, volume overload and heart failure are indistinguishable-clinicians who ignore the tidal nature of volume status in patients who undergo thrice weekly hemodialysis will miss the clinical boat. For this reason, a functional classification scheme for heart failure (specific to dialysis patients) was published in 2014 (8). Administrative data may be even more treacherous, as shown in Figure 2. Based on Medicare claims, heart failure and/or cardiomyopathy hospitalizations decreased dramatically (with a corresponding increase in volume overload hospitalizations) from 2003 to 2012. Although a few (blinded) observers might view this temporal trend in reduction of heart failure events optimistically, Wetmore et al. (9) charitably ascribed it to "coding drift."
|
|
| |
| |
|
|
|