| |
PCSK9 price-cut matchup is on, as Regeneron and Sanofi slash Praluent list tag 60%
|
| |
| |
Feb 11, 2019
https://www.fiercepharma.com/pharma/pcsk9-price-cut-matchup-as-regeneron-and-sanofi-slash-praluent-list-tag-60
It's not often that a tit-for-tat pricing battle goes public in biopharma-and that sort of fight is perhaps even more rare for prerebate list prices.
But Regeneron and Sanofi are engaged in just that with their PCSK9 cholesterol drug rival Amgen. Effective for pharmacy orders in early March, they will slash the list price of their entrant, Praluent, by 60%, matching the $5,850 level Amgen has reached with head-to-head competitor Repatha.
"We hope that payers will do their part to help ensure savings are directly passed on to more patients, through lower out-of-pocket costs," Sanofi's North America head of Primary Care Business Unit, Michelle Carnahan, said in a statement on Monday.
Launched in 2015, both Repatha and Praleunt carried original list prices of a bit more than $14,000 a year. But drug pricing watchdogs at the Institute for Clinical and Economic Review (ICER) argued at the time that the PCSK9 inhibitors needed to bear prices of $5,404 to $7,735 to be cost-effective, drawing counterattack from Amgen and Regeneron.
But in March 2018, Sanofi and Regeneron reported that Praluent reduced the risk of cardiovascular events by 15%, and in high-risk patients with high levels of LDL, the benefit was even greater. Trying to capitalize on those positive results to lock in market share, the pair made a first move toward a dramatically lower price, cutting Praluent's postrebate net price to the range of $4,500 to $8,000-within ICER's recommendation-and won themselves an exclusive spot on Express Scripts' preferred formulary.
Amgen didn't just sit around. Last October, in what may be a first for pharma, Amgen slashed the list price of Repatha by 60% to $5,850 per year. In an interview with FiercePharma at the time, Amgen EVP of global commercial operations Murdo Gordon said the rebates the company was paying to commercial insurers and PBMs didn't lower patients' costs, which many payers peg to a drug's list price. Amgen could only fix that by cutting Repatha's retail price.
RELATED: For sale: Amgen chops 60% off Repatha's $14,520 retail price
Now, Regeneron and Sanofi have followed suit, and the two competitors are once again on the same price level.
"Offering a lower-priced Praluent will help lower seniors' out-of-pocket costs and thereby remove another barrier to receiving this important medicine," Regeneron helmsman Leonard Schleifer said in a statement. The team now estimates that Medicare patients' out-of-pocket costs will drop to about $25 to $150 a month.
Praluent has been lagging behind Repatha in terms of sales. In 2018, it generated sales of 261 million ($295 million), versus the Amgen drug's $550 million. Those numbers represent year-over-year increases of 56% and 72%, respectively, but are still relatively weak compared with analysts' original excitement over the PCSK9 class.
Bernstein analyst Ronny Gal drew a gloomy outlook when Praluent's net price first came down for Express Scripts, calling the deal a "capitulation" that's almost as good as nixing megablockbuster sales potential for PCSK9 drugs.
Gordon, though, expressed optimism. While admitting that lower net prices will take a toll on Repatha numbers in the near term, "we expect to see a positive impact on volume growth and reported net sales over the longer timeframe," the Amgen exec said on the company's fourth-quarter earnings call.
------------------
UnitedHealthcare demands drug rebates even if pharma cuts list prices: analyst
https://www.fiercepharma.com/pharma/letter-to-pharmas-unitedhealthcare-seeks-to-protect-drug-rebates-from-price-reductions
If drugmakers think they can save on rebates if they cut list prices as politicians and public opinion are demanding-well, forget it, says UnitedHealthcare, which sent new demands to pharma companies, an analyst wrote.
The insurance and pharmacy benefits giant is demanding long notices ahead of any drug price cuts, according to the letter, which two drugmakers confirmed to Bernstein analyst Ronny Gal. And UnitedHealthcare expects equivalent rebates whenever list prices are cut, the analyst wrote in a Friday note to investors.
The news comes as drug companies look to price reductions as a new strategy to fight high rebates and gain goodwill with lawmakers and the Trump administration. On Monday, Sanofi announced that it is cutting its Praluent price by 60%, following Amgen's move to chop Repatha's list price by the same percentage. The PCSK9 cholesterol drugs are among many that have a large "gross-to-net" price gap, or high list prices-and high rebates and discounts paid out to the supply chain.
Lowering list prices means smaller costs for patients, but the strategy would also mean lower revenues for PBMs.
UnitedHealthcare asked for seven quarters' notice-a full 21 months-when companies intend to lower prices, Gal wrote. The "drug companies are not too happy about" the UnitedHealthcare letter, he added, as many are considering price reductions.
Gal published another note Monday with UnitedHealthcare's response. The insurance giant's investor relations team reached out to the analyst and said they believed the original report on the letter was misleading. For one, UnitedHealthcare's OptumRx sent the letters in late December and early January, before the administration's recent rebate proposal, Gal wrote, adding that they relate only to rebates in Medicare Part D.
The company explained that Part D contracts "are done on an annual basis and must be submitted to CMS six months ahead of coming into effect," Gal wrote. UnitedHealthcare needs the time to calculate drug cost structures, Gal wrote, summarizing the discussion. And on maintaining rebates, UnitedHealthcare told the analyst patient premiums would rise with lower rebates.
--------------------------
Sanofi and Regeneron cut list price of cholesterol drug by 60 percent
February 11, 2019
https://www.reuters.com/article/us-sanofi-fr-regeneron-cholesterol/sanofi-and-regeneron-cut-list-price-of-cholesterol-drug-by-60-percent-idUSKCN1Q019M
NEW YORK (Reuters) - Sanofi SA and Regeneron Pharmaceuticals Inc said on Monday that they will slash the U.S. list price of their potent but expensive cholesterol fighter Praluent by 60 percent, as the drugmakers follow a similar move by rival Amgen Inc in hopes of increasing use of the drug.
The new list price for Praluent will be $5,850 a year, matching the price Amgen set when it lowered the list of its competing drug, Repatha, in October.
Sanofi and Regeneron said they expect the lower-priced Praluent to be available for pharmacies to order in early March. They said the new price should improve patient access and result in lower out-of-pocket costs for U.S. consumers.
Praluent and Repatha belong a class of injectable biotech drugs called PCSK9 inhibitors that dramatically lower bad LDL cholesterol and reduce the risk of heart attacks and death.
Sales of both have been severely constrained by onerous roadblocks to patient access put up by insurers looking to limit spending on the expensive drugs.
They were approved in 2015 with initial list prices of more than $14,000 a year.
In March of last year, Regeneron and Sanofi said that they would be willing to charge less for their drug if insurers agreed to reduce barriers for high-risk heart patients.
A few months later they struck a deal with Express Scripts, now part of Cigna Corp, to make the drug available to that company's customers at a price in the range of $4,500 to $6,600 a year.
The United States, which leaves drug pricing to market competition, has higher prices than in other developed countries, where governments directly or indirectly control costs. That makes it by far the world's most lucrative market for manufacturers.
Congress has been targeting the pharmaceutical industry over the rising cost of prescription drugs for U.S. consumers, particularly since Democrats took over the House of Representatives in January.
Executives from at least six drugmakers plan to testify at a Senate hearing on rising prescription drug prices later this month.
Drug pricing is also a top priority of the administration of President Donald Trump, who had made it a central issue of the 2016 presidential campaign.
---------------------------
US price of Praluent slashed by 60%
11-02-2019 Print
Joining in what looks set to be a price war for biotech drugs called PCSK9 inhibitors for the treatment of bad cholesterol, USA-based Regeneron Pharmaceuticals (Nasdaq: REGN) and partner French pharma major Sanofi (Euronext SAN) today announced a further reductions of their partnered product.
Praluent (alirocumab) will be made available at a new reduced US list price of $5,850 annually, a 60% reduction from the original price, for both the 75mg and 150mg doses, beginning in early March.
This follows on the news that rival PCSK9 inhibitor Repatha (evolocumab) from Amgen (Nasdaq: AMGN) has its price cut by 60% last October.
Both drugs were approved in 2015 with initial list prices of more than $14,000 a year. Regeneron Praluent sales in full-year 2018 were $306.8 million, up 5.6%, while Repatha generated $550 million revenues.
Aimed to help patient access and affordability
The new lower-priced Praluent is expected to result in lower patient out-of-pocket costs and represents another step in the companies' efforts to help improve patient affordability and access. This follows an earlier announcement in March 2018, when Regeneron and Sanofi committed to lower the US net price for payers in return for helping to reduce burdensome access barriers for appropriate patients.
"In 2018, we lowered the Praluent net price for health plans that were willing to improve patient access and affordability. While lowering the net cost to payers did improve access, seniors who were prescribed Praluent were often still unable to afford it due to high co-pay costs or co-insurance at many Medicare Part D plans," said Dr Leonard Schleifer, president and chief executive of Regeneron. "Offering a lower-priced Praluent will help lower seniors' out-of-pocket costs and thereby remove another barrier to receiving this important medicine."
With the new lower-priced Praluent, most Medicare Part D patients are expected to pay between $25 and $150 per month, a potential savings of up to $345, depending on their insurance plan. Eligible commercial patients will continue to have access to co-pay assistance through MyPraluent.
The new, lower-priced Praluent is expected to be available for pharmacies to order in early March, and the doses with the original list price will remain on the market at least through 2019. The lower-priced Praluent will be identical to the Praluent currently available, other than the list price.
----------------------------
Regeneron, Sanofi cut PCSK9 list price, matching earlier move by rival Amgen
https://www.biopharmadive.com/news/regeneron-sanofi-cut-pcsk9-list-price-matching-earlier-move-by-rival-amge/548147/
Dive Brief:
• Regeneron and Sanofi will sharply cut the list price of their cholesterol medicine Praluent in a rare move designed to reduce patient out-of-pocket costs and spur uptake of the biologic drug, the companies announced Monday.
• Starting in early March, the PCSK9 inhibitor will be available in the U.S. at a list price of $5,850 per year, 60% less than the original price of $14,625 annually. The reduction matches a price cut taken last year by rival Amgen on its competing drug Repatha.
• Both drugs got off to a slow start following U.S. approvals in 2015 as payer pushback on the drugs' similarly high price tags hindered uptake. That both Amgen and the team of Regeneron and Sanofi decided to lower list prices is significant, and could reflect the beginnings of a broader re-evaluation by drugmakers of the role played by drug rebates, particularly in markets with heavy Medicare exposure.
Dive Insight:
Monday's announcement isn't the first effort by Sanofi and Regeneron to trade lower pricing for higher prescription volumes.
Last March, following release of positive cardiovascular outcomes data for Praluent (alirocumab), the companies signaled willingness to reduce the drug's net price after rebates for payers that agreed to drop barriers limiting patient utilization.
Express Scripts, now part of Cigna, was the first to cut a deal, granting exclusive placement on its national formulary in exchange for a new net price then reported to be between $4,500 and $6,000 a year.
By reducing Praluent's price outright, Sanofi and Regeneron have given tacit acknowledgement that the previous offer wasn't sufficient to significantly shift market share.
"In 2018, we lowered the Praluent net price for health plans that were willing to improve patient access and affordability," said Regeneron CEO Leonard Schleifer in a Feb. 11 statement. "While lowering the net cost to payers did improve access, seniors who were prescribed Praluent were often still unable to afford it due to high co-pay costs or co-insurance at many Medicare Part D plans."
Regeneron said most Medicare Part D patients would pay between $25 and $150 per month for lower-priced Praluent, which would represent a savings of as much as $345 according to an illustrative scenario laid out by the biotech.
Coinsurance is often based off a drug's list price, rather than the lower net price reflective of rebates paid by drugmakers to payers. Amgen cited a similar rationale in its move to cut the price of Repatha (evolocumab) last fall.
Recent sales data show Repatha to hold a sizable lead over Praluent in the PCSK9 inhibitor market. Measured by new prescription volume, Repatha has captured a roughly 70% market share, according to Iqvia data cited by Wall Street analysts.
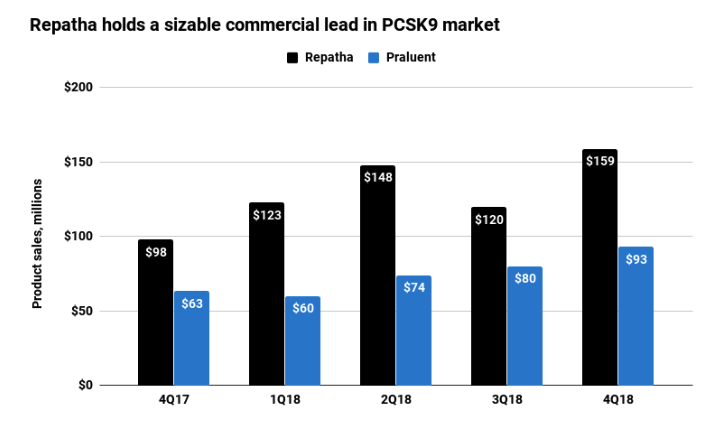
Credit: Ned Pagliarulo / BioPharma Dive, data from companies
Michael Yee, an analyst at Jefferies, recently noted that combined sales of the two drugs are now pacing above a $1 billion per year run rate - a notable threshold for two drugs previously forecast to quickly become blockbusters on their own.
Both drugs are approved to treat certain adults already on maximally tolerated statins who require additional lowering of LDL-C, considered "bad" cholesterol. Repatha is also cleared to reduce the risk of heart attack, stroke or coronary revascularization in adults with established heart disease - an important indication that is seen as having helped Amgen's drug expand its market presence.
For Amgen and the Sanofi/Regeneron team, lowering prices appears to be a bet on higher prescription volumes driving sales higher. In the near-term, however, the drugmakers may see a hit to revenues.
"While the lower net price may impact reported Repatha sales near-term, we expect to see a positive impact on volume growth and reported net sales over the longer timeframe," said Amgen commercial head Murdo Gordon on a recent quarterly earnings call.
|
|
| |
| |
|
|
|