 |
 |
 |
| |
Combination of TMC114/r and TMC125 in Patients with Multidrug Resistant HIV
|
| |
| |
Reported by Jules Levin
J Montaner, M Harris, T Kakuda, G Larsen, B Woodfall, D Miralles, PR Harrigan
Montaner et al., XVI International AIDS Conference, Toronto, Canada, Aug 2006, Poster THPE0136
TMC114 and TMC125 are antiretroviral drugs with activity against HIV that is resistant to available PIs and NNRTIs, respectively. TMC114 is a PI & was approved in June and TMC125 is a NNRTI and is available through an Expanded Access Program from Tibotec.
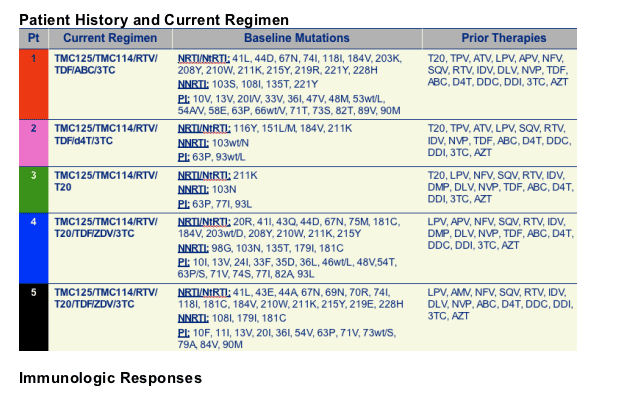
5 Patients
- HIV+ adults
- extensive treatment experience with NRTIs/NtRTIs,
NNRTIs, and PIs
- evidence of drug resistance on genotypic testing
Medication regimens
TMC114 600 mg with ritonavir 100 mg twice daily
TMC125 200 mg twice daily
+/- NRTIs/NtRTIs
+/- enfuvirtide (T20)
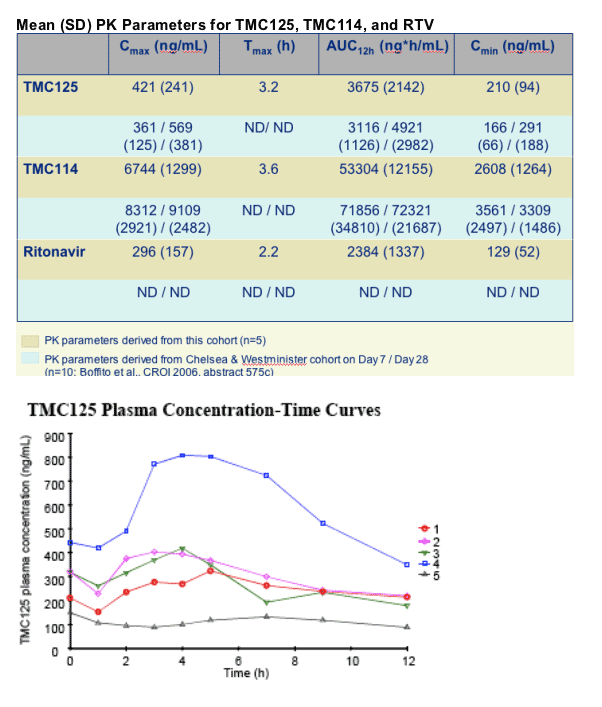
PK analysis: Cmax, Cmin, Tmax, AUC-12h
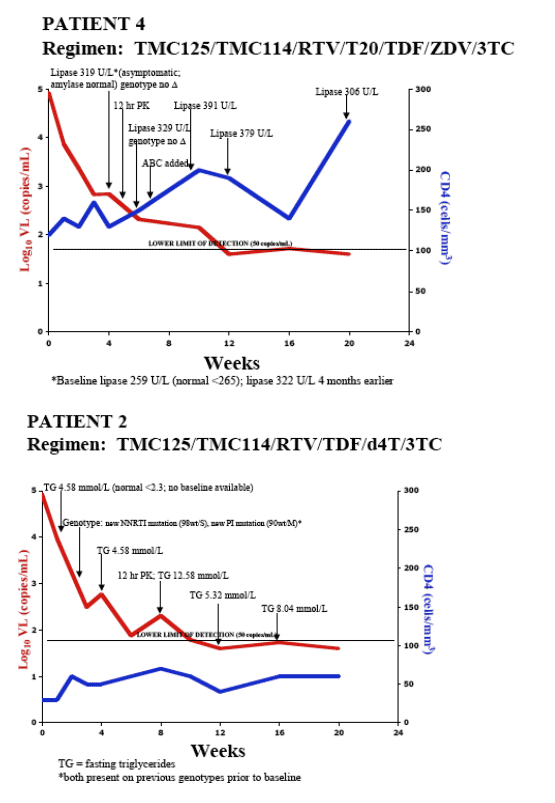
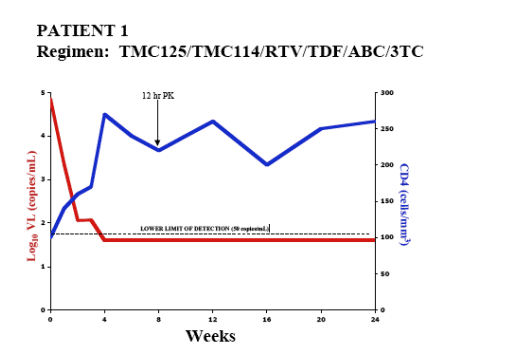
AUTHOR CONCLUSIONS
- In 5 treatment-experienced patients with drug-resistant HIV
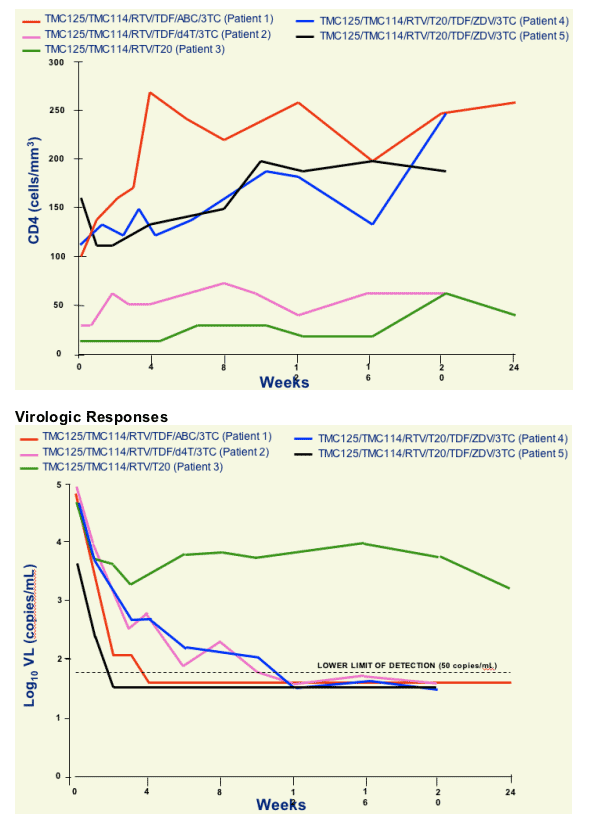
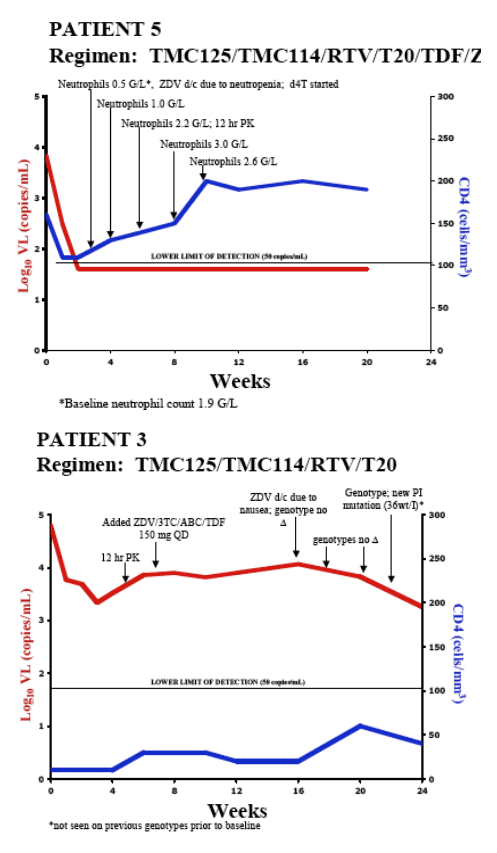
- Combination therapy including TMC114/r and TMC125 resulted in substantial VL declines and CD4 cell count increases over 20-24 wks
- 4/5 achieved HIV RNA <50 copies/mL
- No major safety concerns identified
- TMC114 and TMC125 plasma levels were generally comparable with data previously presented for HIV+ patients
- Combination of TMC114/r and TMC125 may present a viable treatment option for patients with multidrug-resistant HIV
|
| |
|
 |
 |
|
|