 |
 |
 |
| |
Lopinavir/ritonavir as single-drug maintenance therapy in patients with HIV-1 viral suppression: Forty-eight week results of a randomized, controlled, open label, clinical trial (OK04 Study)
|
| |
| |
Arribas JR, Pulido F, Delgado R, Cabrero E, Cepeda C, Gonzalez-Garcia J, Perez-Elias MJ, Arranz A, Portilla J, Pasquau J, Iribarren JA, Rubio R, Ocampo A, Miralles P, Knobel H, Gaya F, Munoz R, Norton M, for the OK04 Study Group
Thursday Late Breaker Oral Presentation at AIDS2006 #THLB0203
Reported by Jules Levin
XVI Intl AIDS Conference, Toronto, Aug 17, 2006
Notes from Toronto. As the World AIDS Conference slows to an end today with closing ceremonies a few thoughts, fond and not so fond. HIV is a unique disease. It stands alone. Twenty-five years ago advocates turned medicine upside down. HIV is a different paradigm in so many ways than any other disease. More patients I think with HIV make their own treatment decisions. Patients are often called advocates. Development of treatments and making HIV a chronic disease has happened so quickly, its unprecedented in medical history. HIV is now a worldwide event. At this conference there were literally hundreds of booths from every region and country you can imagine in the 'Global Village'. HIV+ individuals and advocates worldwide gather at this event, this gathering from all corners of the world, to be a part of their brethren. There was a condom clothing outfit being stared at by a women from Saudi Arabia completely covered only showing her eyes. The initial attendance count was 25,000 at the outset of the meeting but I heard there were 32,000 after a few days. As I have said before, I have never attended such a poorly organized and run conference. Yes it was big but so many poor decisions were made. For the first few days HIV+ individuals had to wait for hours on line to register. It took 25 minutes to walk from the entrance of the conference center to the South Building where posters and oral sessions took place. The big treatment oral Late Breaker on Thursday was in a room the size of an aircraft hangar and there were an unprecedented 18 oral presentations, many of which were very interesting. Seeing Pres Clinton speak was inspiring for many at the conference. I think his job as President was preparation for his current work, which will be what he is remembered for as he plans his legascy. It's always a letdown when a conference ends and it's time to go home. But I just returned to the Big Apple and I'm glad to be home in smelly grimy NYC.
OK04 trial design
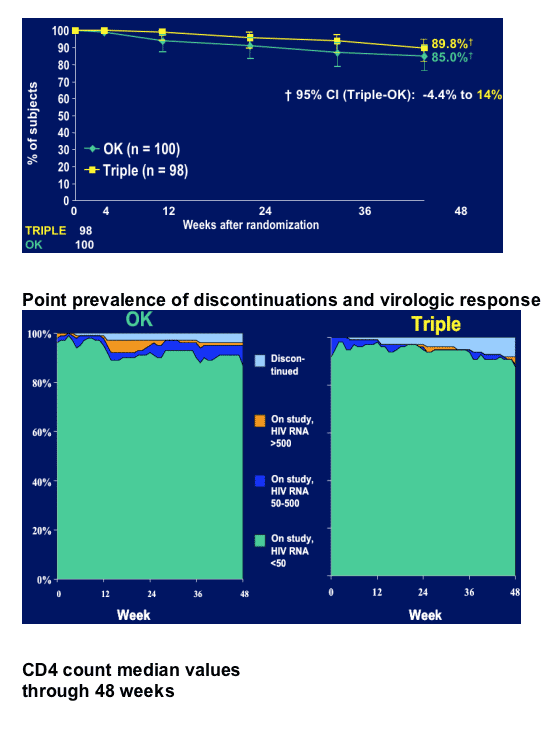
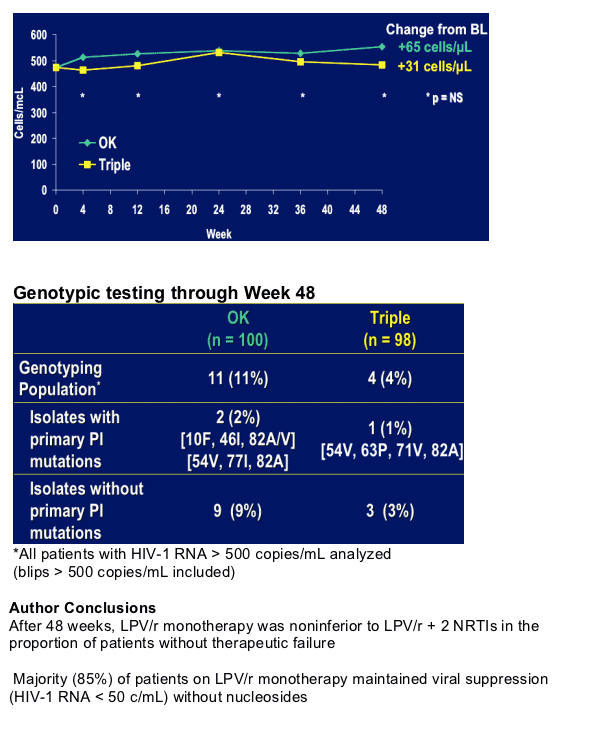
After >6 months with HIV <50 c/ml, no history of PI failure patients received LPV/r soft-gel capsules 400/100 mg bid (n=100), or LPV/r sofa-gel capsules + 2 NRTIs (n=100). This trial is different than the other Kaletra monotherapy studies because reinduction of nubes and resupresion of HIV RNA is NOT considered failure, but is counted as a success in this study.
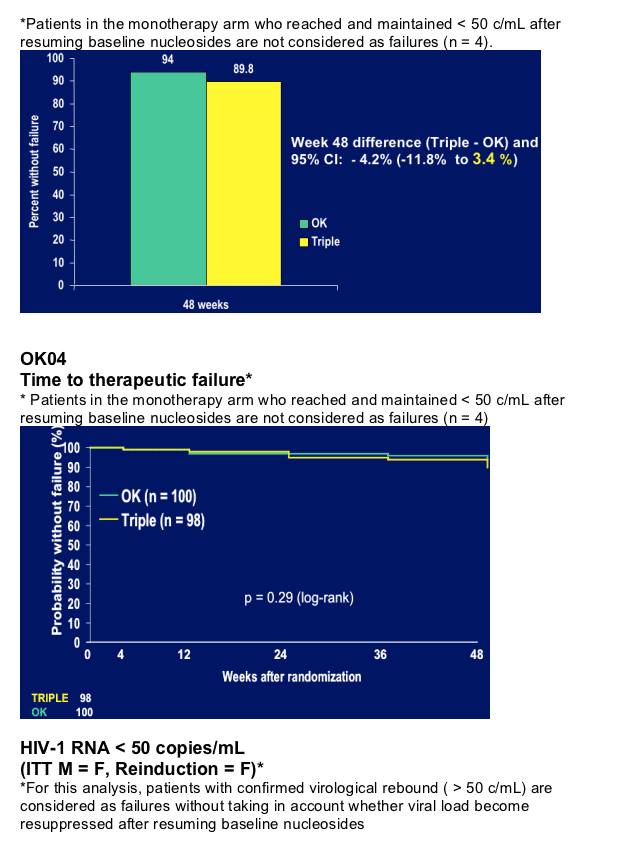
Primary endpoint: Therapeutic failure at 48 weeks
- 2 viral loads > 500 c/mL 2 weeks apart* (without virological re-
suppression after reinduction with NRTI in the OK arm) OR
- Change of randomized therapy for reasons different from re-induction OR
- Treatment discontinuation OR
- Lost to follow-up
*OR decrease in HIV-1 RNA < 1 log 4 weeks after intensification OR
failure to reach HIV-1 RNA < 50 c/mL 16 weeks after intensification
OK04 Trail Design
Primary endpoint
--Proportion of patients without therapeutic failure at 48 weeks
Study power
With 100 patients per arm, study has an 80% power to show the non-inferiority of the OK arm, assuming:
--10% therapeutic failure rate at 48 weeks in both arms
--Non-inferiority would be demonstrated if the upper limit of
the 95% CI of the difference in percent of patients without therapeutic failure (Triple - OK) is < 12%
Randomization stratification
--Nadir CD4 cell count (> or < 200 cells/mL)
--Months HIV-1 RNA < 50 c/mL prior to randomization (> or < 9 months)

|
| |
|
 |
 |
|
|