 |
 |
 |
| |
TMC114/r in treatment-experienced patients in POWER 1, 2 and 3:
integrated analysis of laboratory parameters
|
| |
| |
Reported by Jules Levin
XVI Toronto, Aug 2006
Vangeneugden T,1 Van Baelen B,1 De Paepe E,1 Tomaka F,2 Parys W,1 Miralles D,1 Lefebvre E2
1Tibotec BVBA, Mechelen, Belgium; 2Tibotec Inc., Yardley, PA, USA
poster TUPE0063
ABSTRACT
Background: The protease inhibitor (PI), TMC114 (darunavir), demonstrated potent antiviral activity with low-dose ritonavir (TMC114/r) among treatment-experienced patients in POWER 1 and 2 (TMC114-C213 and C202), and showed similar efficacy in the non-controlled POWER 3 (TMC114-C215/C208) analysis. This analysis assesses relevant laboratory parameters for all patients receiving the recommended dose of TMC114/r 600/100mg bid in POWER 1- 3.
Methods: Patients were PI-, NRTI- and NNRTI-experienced with 31 primary PI mutation at baseline (BL). The original safety analysis was expanded to include all patients who initiated treatment with TMC114/r 600/100mg bid (n=458) in POWER 1- 3 and those who received control PI (CPI) in POWER 1 and 2 (n=124). Given that patients on a failing CPI regimen were allowed to roll over to a comparison study beyond Week 12, analysis at Week 12 provided the most complete comparison and is presented.
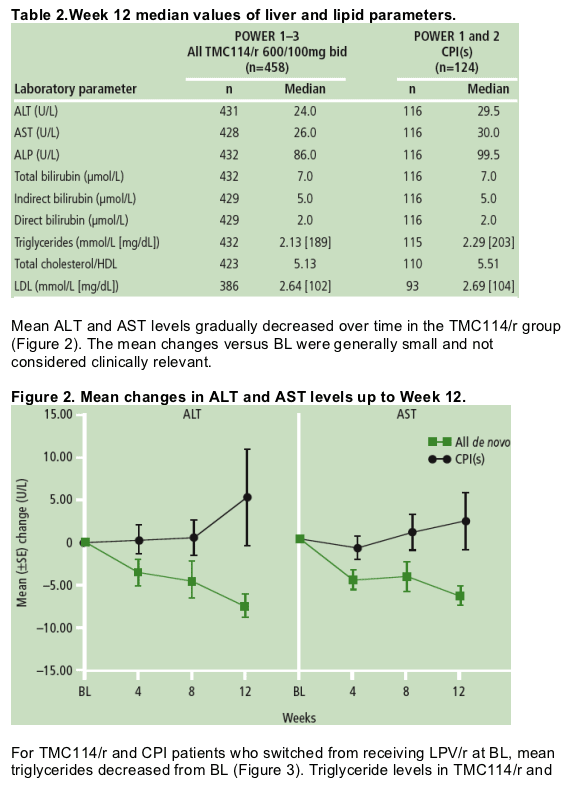
Results: Overall incidence of graded and non-graded laboratory abnormalities was similar across treatment groups. The majority of abnormalities were grade 1- 2 in severity and discontinuations were infrequent. Median values of liver and lipid parameters are shown in the Table.
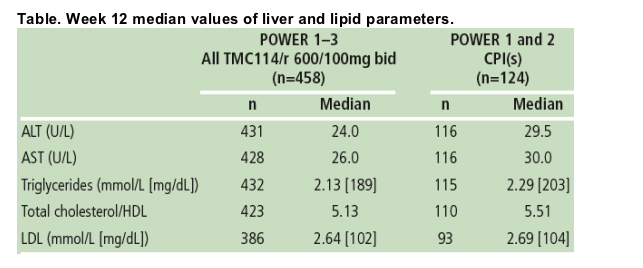
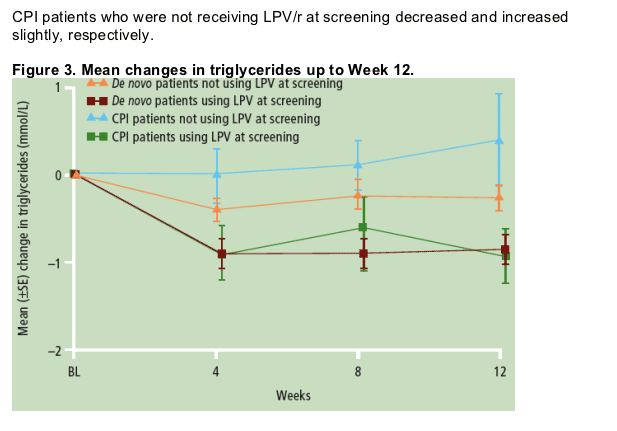
Mean triglycerides decreased both for TMC114/r and CPI treatment for patients who switched from lopinavir with low-dose ritonavir (LPV/r) at BL.
Conclusions: No organ-specific toxicological signal was observed with TMC114/r. These data suggest that routine follow-up for patients receiving TMC114/r is adequate. Ongoing phase III studies will provide more data on the safety of TMC114/r.
The analysis included all patients who initiated treatment with TMC114/r 600/100mg bid (de novo) plus an investigator-selected optimized background regimen (OBR, consisting of >/=2 NRTIs with or without enfuvirtide [ENF]) in POWER 1, 2 and 3 and those who received CPI(s) plus OBR in POWER 1 and 2.
A total of 582 patients were included in the analysis, of which 458 initiated treatment with TMC114/r 600/100mg bid plus OBR in POWER 1, 2 and 3, and 124 received CPI(s) plus OBR in POWER 1 and 2.
The overall rate of treatment discontinuation was higher with CPI than with TMC114/r treatment (81% vs 11%). The discontinuation rate due to virologic failure was higher in the CPI group (67% vs 3% in the TMC114/r group) but the rate due to an AE or HIV-related event was comparable between the two groups (5% for CPI and 4% for TMC114/r).
BL characteristics
- BL characteristics were similar between the two treatment groups (Table 1).
- The investigator-selected PIs used in the CPI group were LPV (36%), saquinavir (35%), amprenavir or fosamprenavir (34%), atazanavir (17%), indinavir (2%) and nelfinavir (1%). Seventy-five percent of regimens were single-boosted PIs, 23% were double-boosted PIs and 2% were double PI combinations without low-dose RTV. Of the 124 CPI-treated patients, 98% were on an RTV-boosted regimen.
Evaluation of laboratory parameters
- The overall incidence of graded and non-graded laboratory abnormalities was generally low and similar between TMC114/r- and CPI-treated patients.
- The majority of abnormalities were grade 1- 2 in severity and discontinuations due to laboratory abnormalities were infrequent (1% for both TMC114/r- and CPI-treated patients).
- The most common grade 3 or 4 laboratory abnormalities in the TMC114/r group were decreased white blood cell (WBC) count (6% vs 7% in the CPI group), and increased triglycerides (9%), amylase (7%) and total cholesterol (5%) (compared with 7%, 5% and 2%, respectively, in the CPI group).
- For the incidence of grade 3 or 4 hematologic abnormalities other than WBC count, TMC114/r- and CPI-treated patients had the same rates for neutrophils (5%), plasma prothrombin time (1%) and platelet count (1%). TMC114/r was associated with higher rates than CPI for partial thromboplastin time (4% vs 2%) and hemoglobin (1% vs 0). On the other hand, TMC114/r had lower rates than CPI for total absolute neutrophil count (4% vs 5%) and lymphocytes (4% vs 10%).
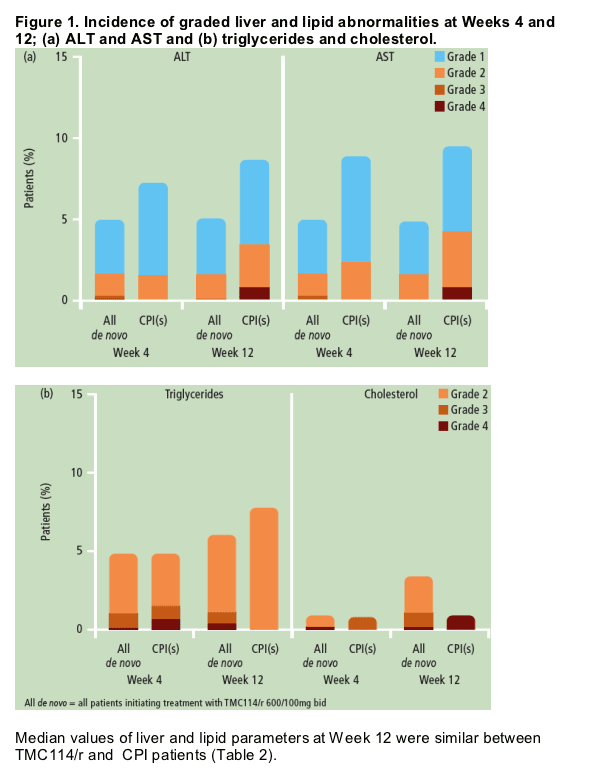
- The incidence of graded ALT, AST, triglyceride and cholesterol abnormalities at Weeks 4 and 12 are shown in Figure 1.
Author Conclusions
- No organ-specific toxicological signal was observed with TMC114/r treatment.
- most laboratory abnormalities were grade 1 or 2 in severity
-- discontinuation due to laboratory abnormalities was <1%.
- Results of this analysis suggest that routine follow-up for patients receiving TMC114/r is adequate.
- Ongoing phase III studies in treatment-naïve (ARTEMIS [TMC114-C211]) and
treatment-experienced patients (TITAN [TMC114-C214]) will provide more data on the safety of TMC114/r.
|
| |
|
 |
 |
|
|