 |
 |
 |
| |
Does Treating Primary HIV-Infection Do Any Good?
|
| |
| |
A report from the XVI International AIDS Conference
Toronto, August 13-18, 2006
Mark Mascolini
Not so long ago--around the time of the 1998 World AIDS Conference in Geneva--treating primary infection seemed a scintillant idea. At that gathering Harvard's Bruce Walker espoused the idea of stopping viral replication with hair-trigger treatment in just-infected people--before HIV wiped out too many of the CD4s programmed to kill it. With those HIV-specific CD4 troops intact, he proposed, people treated during primary infection might be able to "inoculate" themselves with their own virus during drug breaks and eventually stop treatment altogether and let the immune system muffle HIV for a lifetime.
Everyone knows that plan came up short in the pilot trial Walker ran in Boston [1]. At the 2002 International AIDS Conference, Spanish clinicians testing the same idea reported similarly poor results [2]. And at this year's international meeting, a French team delivered what may be strike three for this tactic with long-term follow-up of the PRIMSTOP trial, detailed below [3].
Even if this start-and-stop strategy does not allow enduring HIV control without drugs, many suggest other cogent reasons for harnessing HIV when people get diagnosed soon after infection--beginning with the commonsensical concept that it can't be bad to stop a ruthlessly regenerative virus whose paths remain poorly understood. And some of the paths that have been plotted argue for swift treatment. Recent research in monkeys, for example, shows that untreated primary HIV infection quickly kills about half of their total stock of memory CD4 cells [4,5].
Despite such compelling evidence, proving the long-term or even medium-term value of prompt treatment remains tough, as two studies unveiled at the AIDS Conference and two just-published reports show. But first the long-term PRIMSTOP findings.
PRIMSTOP doesn't stop HIV for long
PRIMSTOP researchers reached the bleak conclusion that "virtually no patient" who took structured drug breaks after treatment for primary HIV infection managed to keep the virus under wraps without antiretrovirals [3]. Depending on how one defines viral control, they may have done without that "virtually."
Like two other pilot trials of drug breaks after brisk treatment [1,2], PRIMSTOP was not randomized because most people want to make early-treatment decisions themselves and most review boards fret over the ethics of withholding treatment. Bruno Hoen and colleagues throughout France signed up 29 people who started antiretroviral therapy during symptomatic acute HIV infection and kept taking their drugs for 34 weeks. At that point, with sub-50-copy viral loads, they all began an interruption scheme of 2, 4, and 8 weeks off therapy interleaved with 12-weeks treatment stints.
At study week 84 the 26 people still in the trial stopped treatment again to see how many could make it to week 108 with a sub-50 load, which was PRIMSTOP's primary endpoint. Only 1 of 26 (4%) did. But 3 (11.5%) managed to stay under 400 copies to week 108, and 5 (19%) kept their load under 1000.
Hoen and coworkers invited these 26 people to keep coming back for checkups, and 22 agreed. Six of the 22 (27%) had to resume antiretrovirals after a median 12 months following PRIMSTOP week 84. One of them was the solitary person whose viral load remained undetectable during weeks 84 to 108. This person restarted antiretrovirals with a viral load still below 10,000 copies but with a chancy CD4 count of 248.
For this PRIMSTOP participant, seemingly seamless early viral control translated into 25 months without therapy. The other 5 had to return to treatment even sooner, after 7, 11, 11, 13, and 18 months off therapy, with CD4 counts ranging from 197 to 266 and with viral loads ranging from 4.18 to 5.44 log copies (about 63,000 to 275,000 copies).
Sixteen people (73%) had not resumed treatment for 30 months after stopping at PRIMSTOP week 84 through the last follow-up before this report. At that point their CD4 counts ranged from 333 to 639 (median 456), a median loss of 240 CD4 cells. During this extended drug break, viral loads in 11 of these 16 people (69%) topped 10,000 copies. Two of the 16 (12.5%) had a clinical reversal during this time, one with oral candidiasis and one with the considerably more ominous diagnosis of non-Hodgkin lymphoma.
For the 14 people (64%) who managed to stay off treatment at least 30 months after the last PRIMSTOP drug break without a clinical setback, the question is whether they would have done worse if they'd never started antiretrovirals in the first place. German physicians tried to answer that question by comparing the value of starting treatment early--then stopping--with avoiding treatment entirely until the need arose:
Start early to stop for a while?
German clinicians offered an unorthodox comparison of two primary infection cohorts to see if they could smoke out an advantage in immediate treatment of early HIV infection [6]. The analysis involved two 100-person cohorts, the first of which began enrollment in July 2001 to look at speedy treatment of primary infection, the second of which began signing people up in January 2003 to assess delaying treatment of primary infection. As it turned out, though, 144 people started antiretrovirals, while 56 did not.
As one would expect, people who opted for immediate treatment had higher viral loads and lower CD4 counts than people who decided to wait. The median first viral load stood above 500,000 copies in the treated group versus a still-high 240,000 copies in the untreated group, a highly significant difference (P < 0.001). Median first CD4 counts were 453 (range 120 to 1342) in the treated group and 621 (range 266 to 1543) in the untreated group, also a significant difference (P = 0.0017). Median CD4% also proved lower in the treated group (24% versus 29%, P = 0.053).
Comparing people who started antiretrovirals immediately and later stopped with people who delayed treatment entirely, the clinicians wanted to see how long it would take each group to reach a standard signal for initial therapy--a CD4 count under 350 or a viral load above 100,000 copies.
Ninety-eight of 144 people (68%) who began antiretrovirals during primary infection stopped after a median 9.0 months (range 1.2 to 28.7 months). In that group 37 (38%) reached the benchmark of fewer than 350 CD4s or more than 100,000 copies a median of 14.3 months after they suspended therapy. In contrast 20 of 56 originally untreated people (36%) reached one or both of those benchmarks a median of 8.3 months after HIV seroconversion. This difference reached statistical significance (P = 0.02) only for people with a first viral load above 50,000 copies/mL.
The critical question is when a person with early infection wants to embrace the benefit and burden of antiretroviral therapy. The early-treatment group had to take antiretrovirals for 9 months on average to enjoy a 14-month break in therapy before their CD4s or viral load told them to start again. The deferred-treatment group saw the start-therapy semaphore (signal) only 8 months after primary HIV infection, so they reached CD4 or viral load signals to start treatment faster than the early-treatment group reached signals telling them to resume treatment. But the deferred-treatment group didn't take antiretrovirals for 9 months to get to the starting line of this comparison.
And one should not overlook the finding that 45 of 144 people who started early therapy--almost one third of that group--decided to play it safe and keep taking their antiretrovirals.
Two published studies pondering the same question
Two similar cohort comparisons published just after the International AIDS Conference tried to reckon whether therapy during acute or early HIV infection leaves people with a lower viral load or higher CD4 count than they could expect if they did not start swallowing antiretrovirals right away. Again, these studies are not randomized trials, so confounding biases may taint the results.
The larger of these two analyses compared viral loads in 13 people who began treatment within 2 weeks of acute infection (the "acute" group), 45 who started 2 weeks to 6 months after acute infection (the "early" group), and 337 people who declined treatment for acute or early infection [7]. The acute group kept taking antiretrovirals for a median of 81 weeks (range 13 to 173 weeks), while the early group stayed on treatment for a median of 70 weeks (range 13 to 260). Frederick Hecht (University of California, San Francisco) and colleagues at other centers began tracking their viral loads and CD4 counts from 4 weeks after they stopped therapy.
Initial viral loads were higher and CD4 counts lower in the two treated groups than in the 337 untreated people, but differences between groups were not statistically significant. Compared with the untreated people, whose viral loads and CD4s were measured from enrollment in the cohort, the acute group had significantly lower viral loads at 24 weeks in an unadjusted analysis and in an analysis adjusted for baseline viral load and CD4 count (Table). At 48 weeks and 72 weeks the viral load difference between the acute group and the untreated group remained statistically significant in the adjusted analysis but not in the unadjusted analysis. Compared with the untreated group, the early group had a significantly lower viral load at 24 weeks in both unadjusted and adjusted analyses (Table). But they lost that advantage in both analyses by week 48.
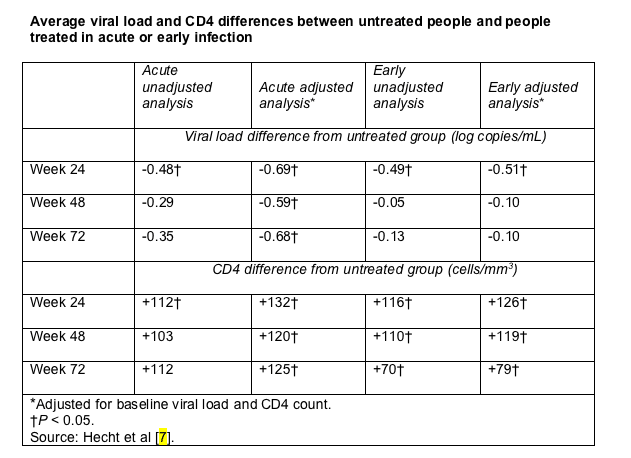
In adjusted analyses both the acute and early groups maintained a significantly higher CD4 count at week 72. But in an unadjusted analysis the acute group did not have a significant CD4 advantage over the untreated group after week 24.
Hecht and colleagues caution that the adjusted analyses "may overstate
the benefits of treatment during acute HIV infection" for the reasons explained in note 8.
Testing a much shorter spell of treatment for acute infection--24 weeks--a team of German and US researchers found no viral load or CD4 benefit 24 weeks after therapy stopped compared with people who did not take antiretrovirals for acute infection [9]. This study involved 20 people who sought care for acute retroviral syndrome in a Berlin clinic, where they had a chance to begin antiretrovirals or to put off treatment. The 12 people who elected to begin therapy did not differ significantly from the untreated people in viral load, CD4 count, clinical symptoms, or variables that may affect early progression of HIV infection.
The 12 treated people all reached a viral load below 50 copies, and all had a viral rebound when they stopped treatment. Six months later the treated group did not differ from the untreated group in viral load or CD4 count, even though when they stopped therapy the treated people had higher levels of CD8 cells that target HIV.
The authors of this small study conclude that "several immunological markers that are currently used to characterize HIV-1-specific CD8+ T cell responses fail to predict the ability of the immune system to control viral replication." They did not take colon biopsies to see whether immediate antiretroviral therapy affects viral onslaughts there, but a team reporting at the International AIDS Conference did:
Fast treatment does not protect CD4s in gut
Shielding critical CD4 cells from early annihilation by HIV remains an attractive rationale for rapid treatment of early infection. But a study of two groups promptly treated at New York's Aaron Diamond AIDS Research Center found that fast treatment that continues for years did not protect gut T cells from HIV or restore them to normal levels [10].
With colleagues at other site, Saurabh Mehandru studied two groups:
- Eighteen people with acute or early HIV infection followed prospectively during 3 years of antiretroviral therapy.
- Twenty-two people diagnosed with acute or early HIV infection and followed after 1, 1 to 3, or 3 to 7 years of treatment.
Mehandru compared rectosigmoid colon biopsies from these people with biopsies from 18 people without HIV infection. Whereas the 18 uninfected people had equivalent CD4 tallies in peripheral blood and colon, the HIV-infected groups had persistently lower CD4 counts in the colon regardless of how long they took antiretrovirals. These results mirror findings of another study published during the Toronto meeting [11].
The Aaron Diamond study also disclosed persistent depletion of CD4 cells expressing the CCR5 coreceptor in promptly treated people and disproportionately high levels of activated CD4 and CD8 cells. Those findings, the researchers suggested, could point to "ongoing viral replication in the gastrointestinal tract during therapy," speculation buttressed by another recent study [12].
Randomized results on the way
The studies reviewed above are hardly the only ones that address the potential merits of unleashing antiretrovirals in the first weeks of infection. Analyzing the two published studies [7,9], Sabine Kinloch-de Loes (Royal Free Centre for HIV Medicine, London) reviews other pertinent trials of the past 5 years in a free online article [13]. Prudently, she suggests no firm conclusions from work presented to date, and such conclusions will remain unwarranted until randomized trials reach fruition. Kinloch-de Loes ran the first randomized trial of treatment for primary infection--with AZT monotherapy [14]. Two trials of more recent regimens are under way (see note 15).
References and Notes
1. Kaufmann DE, Lichterfeld M, Altfeld M, et al. Limited durability of viral control following treated acute HIV infection. PLoS Med. 2004;1(2):e36 (http://www.plosmedicine.org/perlserv/?request=get-document&doi=10.1371/journal.pmed.0010036).
2. Miro JM, Plana M, Garcia F, et al. Structured treatment interruption in patients receiving HAART within 90 days after onset of primary HIV-1 infection symptoms: spontaneous control of viremia in only one third of cases after four cycles off therapy. 14th International AIDS Conference. July 7-12, 2002. Barcelona Abstract ThOrB1437.
3. Hoen B, Deveau C, Fournier I, et al. Absence of sustained benefit of HAART followed by structured treatment interruptions in primary HIV-1 infection: prolonged follow-up of patients enrolled in the PRIMSTOP (ANRS 100) trial. XVI International AIDS Conference. August 13-18, 2006. Toronto. Abstract MOPE0059.
4. Li Q, Duan L, Estes JD, et al. Peak SIV replication in resting memory CD4+ T cells depletes gut lamina propria CD4+ T cells. Nature 2005;434:1148-1152.
5. Mattapallil JJ, Douek DC, Hill B, et al. Massive infection and loss of memory CD4+ T cells in multiple tissues during acute SIV infection. Nature 2005;434:1093-1097.
6. Koegl C, Wolf E, Jessen H, et al. Does early treatment of primary HIV infection delay treatment indication? XVI International AIDS Conference. August 13-18, 2006. Toronto. Abstract MOPE0060.
7. Hecht FM, Wang L, Collier A, et al. A multicenter observational study of the potential benefits of initiating combination antiretroviral therapy during acute HIV infection. J Infect Dis 2006;194:725-733 (http://www.journals.uchicago.edu/JID/journal/issues/v194n6/35358/35358.html).
8. "Because study subjects typically decided whether to start receiving ART after they knew their viral load and CD4+ T cell count, the untreated group is likely to be enriched for persons who rapidly achieved low viral loads and maintained good CD4+ T cell counts. Although our statistical adjustment will account for this selection bias, it is likely
to overcorrect for higher viral loads that are associated with earlier stages of acute HIV infection. Thus, the unadjusted analyses are more conservative, and the adjusted analyses may overstate the benefits of treatment during acute HIV infection" (Hecht et al [7]).
9. Streeck H, Jessen H, Alter G, et al. Immunological and virological impact of highly active antiretroviral therapy initiated during acute HIV-1 infection. J Infect Dis 2006;194:734-739 (http://www.journals.uchicago.edu/JID/journal/issues/v194n6/35965/35965.html).
10. Mehandru S, Poles M, Tenner-Racz K, et al. Gastrointestinal immune reconstitution is delayed and incomplete after initiation of antiretroviral therapy during acute HIV-1 infection. XVI International AIDS Conference. August 13-18, 2006. Toronto. Abstract TUPE0033.
11. Guadalupe M, Sankaran S, George M, et al. Viral suppression and immune restoration in the gastrointestinal mucosa of human immunodeficiency virus type 1-infected patients initiating therapy during primary or chronic infection. J Virol 2006;80:8236-8247.
12. Poles MA, Boscardin WJ, Elliott J, et al. Lack of decay of HIV-1 in gut-associated lymphoid tissue reservoirs in maximally suppressed individuals. JAIDS 2006;43:65-68.
13. Kinloch-de Loes S. Treatment of acute HIV-1 infection: is it coming of age? J Infect Dis 2006;194:721-724 (http://www.journals.uchicago.edu/JID/journal/issues/v194n6/36892/36892.html).
14. KinlochĐde Loes S, Hirschel BJ, Hoen B, et al. A controlled trial of zidovudine in primary human immunodeficiency virus infection. N Engl J Med 1995;333:408-413.
15. CTN214 is a Canadian trial randomizing people with acute or early HIV infection to 1 year of therapy or to no therapy (http://www.hivnet.ubc.ca/e/clinicaltrials/A214.html). A study run by the US National Institute of Allergy and Infectious Diseases will randomize people with primary HIV infection to immediate steady therapy or to immediate therapy followed by a treatment break (http://www.clinicaltrials.gov/ct/show/NCT00084032).
|
| |
|
 |
 |
|
|