 |
 |
 |
| |
Pharmacokinetics and Drug Interaction Profile of
Cobicistat boosted-Elvitegravir with Atazanavir, Rosuvastatin or Rifabutin
|
| |
| |
Reported by Jules Levin
13th HIV Clinical Pharmacology Wkp
Apr 16-18 2012
Barcelona Spain
S Ramanathan, H Wang, T Stondell, A Cheng, and BP Kearney
Gilead Sciences, Inc., Foster City, CA, USA

EXCERPTED from:
Clinical Pharmacology at the 13th Workshop on Clinical Pharmacology of HIV Therapy - Courtney V. Fletcher, Pharm.D. - (05/14/12)
------Elvitegravir+Cobicistat drug interactions with rosuvastatin, atazanavir and rifabutin: OK, Not Yet and Not Recommended.
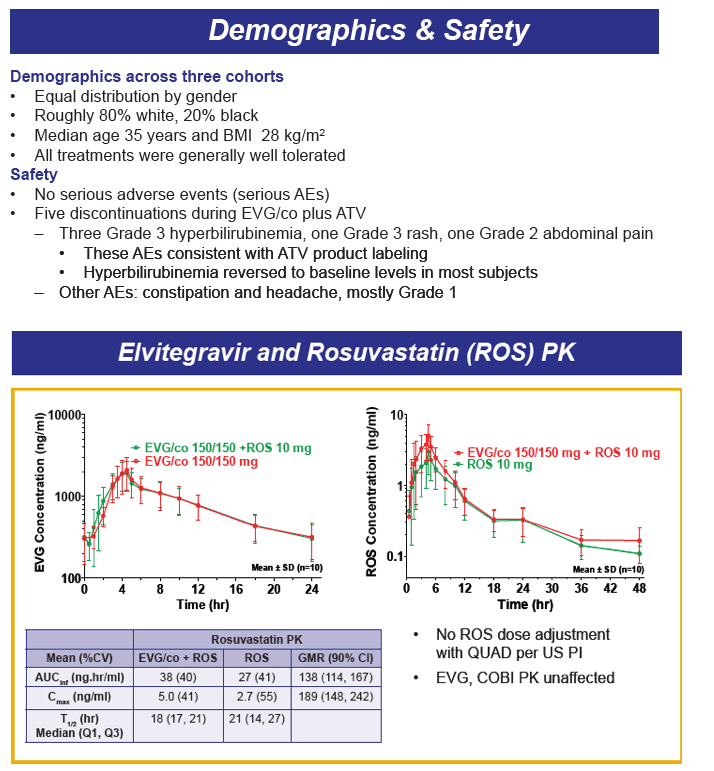
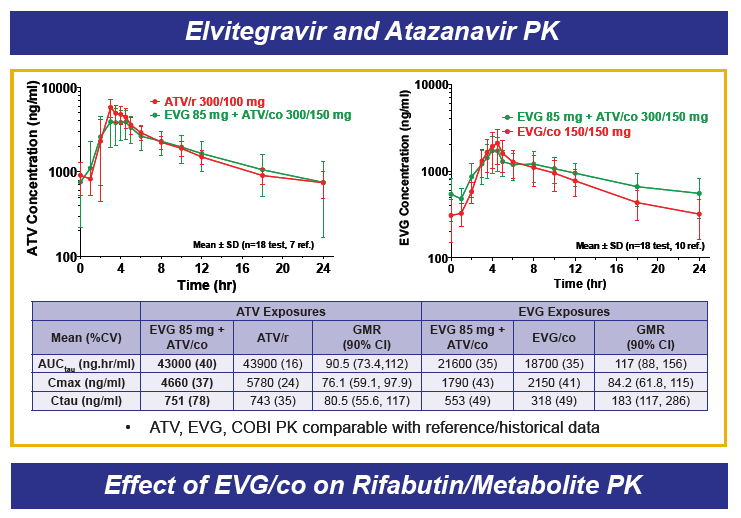
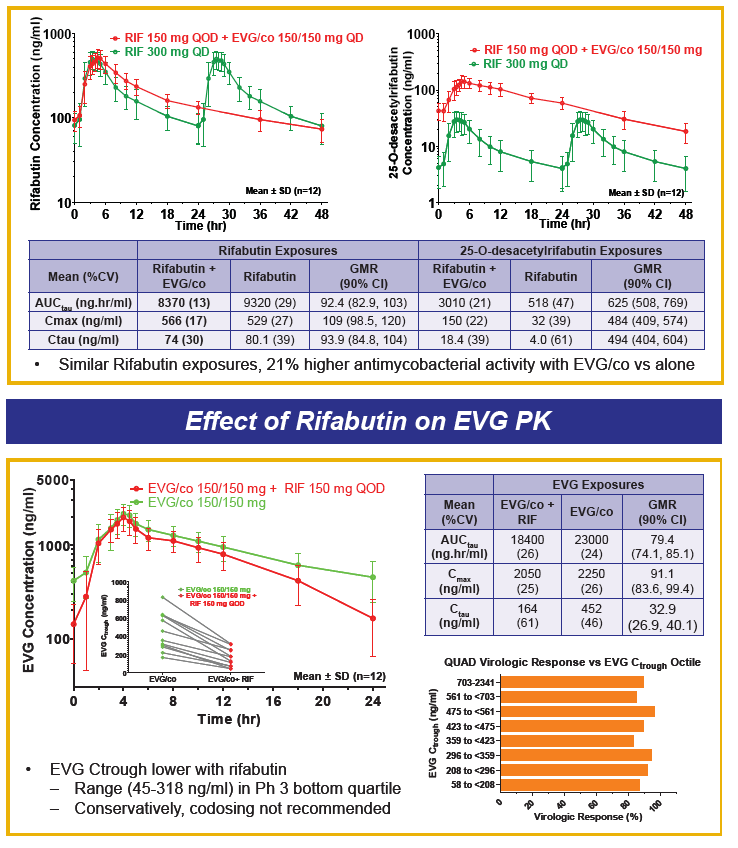
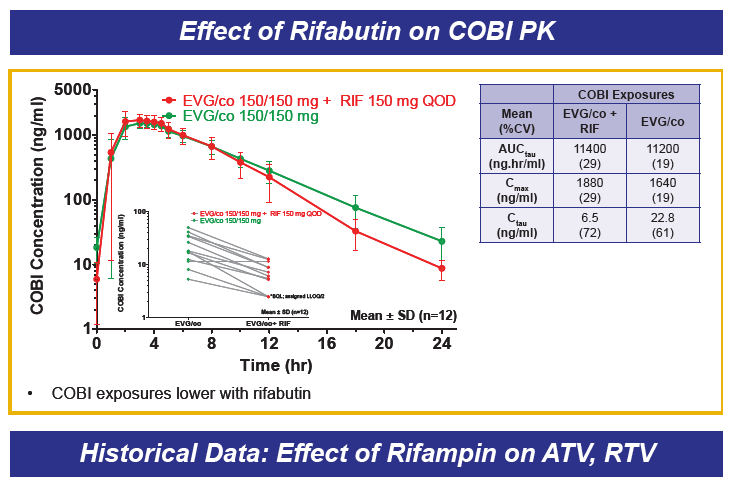
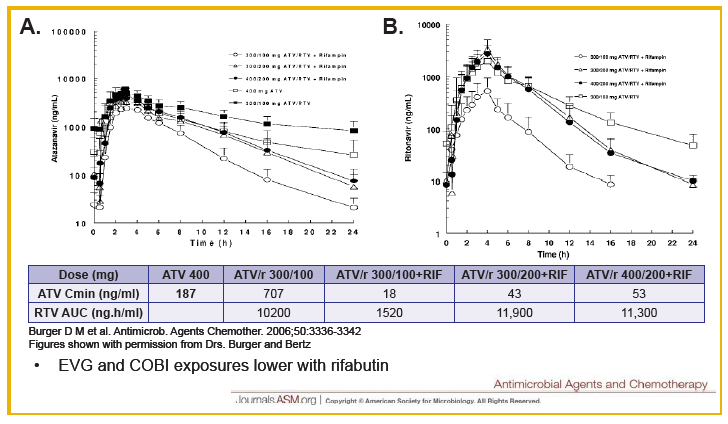
Drug-drug interactions between EVG+COBI and rosuvastatin, atazanavir and rifabutin were evaluated in 3 cohorts of healthy volunteers. The rosuvastatin dose was 10 mg and that for EVG+COBI was 150/150mg. The ATV dose was 300 mg, but the EVG+COBI doses were 85/150mg. The reduced EVG dose is because EVG is metabolized by glucuronidation and ATV is an inhibitor of glucuronidation. The RBT dose was 150 mg every other day when given with EVG+COBI 150/150mg.

Administration of EVG+COBI and rosuvastatin at the usual starting dose of 10 mg/day looks fine. Because rosuvastatin concentrations are increased, however, clinicians should monitor for adverse effects, primarily rhabdomyolysis and liver enzyme elevations especially with use of higher rosuvastatin doses. I think there is work left to do prior to recommending the use of ATV with EVG+COBI. ATV trough concentrations are decreased 20% and the variability in trough concentrations is doubled, when compared with ATV/RTV. There are no data that I am aware of for the combination of ATV with EVG+COBI and + TDF, and the potential of the addition of TDF to decrease ATV trough concentrations even further. So, my conclusion is not yet to an ATV with EVG+COBI combination. Finally, RBT substantially decreases both EVG and COBI concentrations, and concomitant administration is not recommended; this recommendation will also apply to rifampin and rifapentine, thus EVG+COBI will not be an option for the HIV-TB coinfected person requiring rifamycin therapy. ----------

References
1. DeJesus E, et al. JAIDS 43(1):1-5, 2006.
2. Sax et al. CROI, 2012.
3. DeJesus et al. CROI, 2012.
4. German et al. IWCPHT, 2008.
|
| |
|
 |
 |
|
|