 |
 |
 |
| |
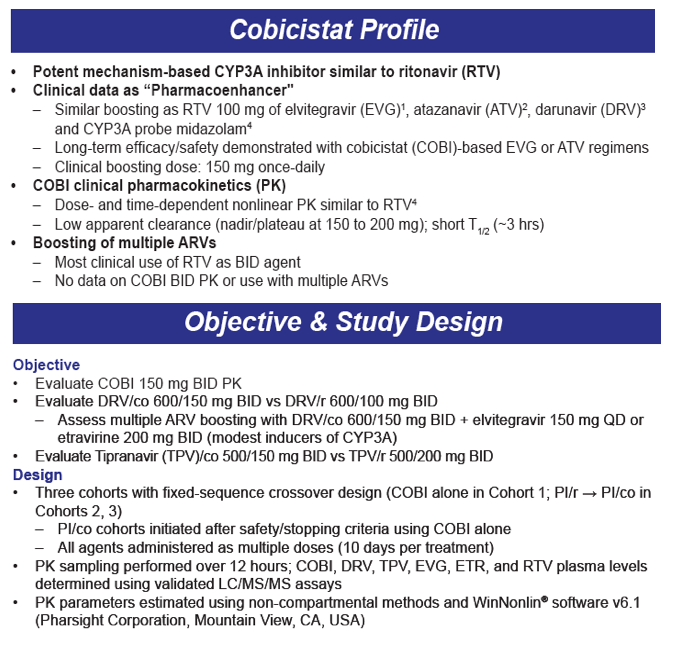
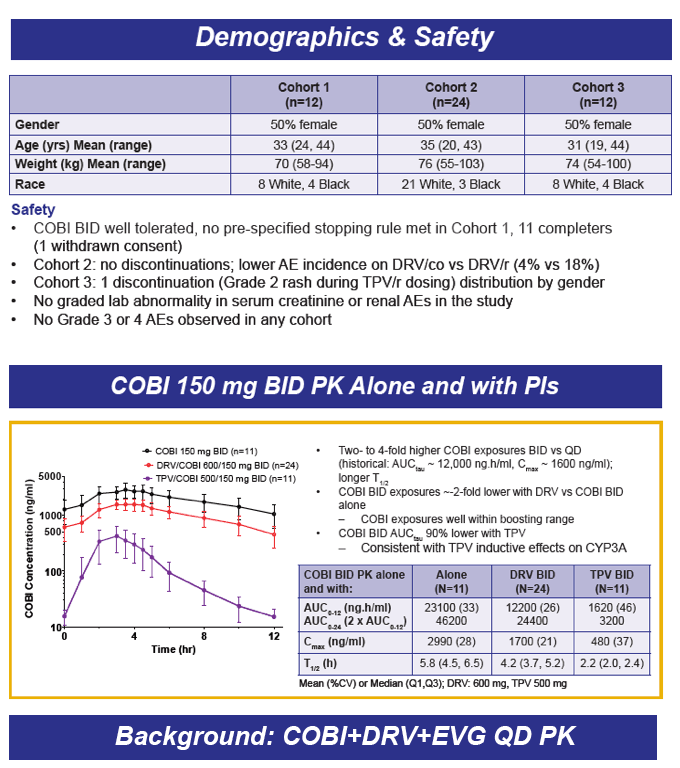
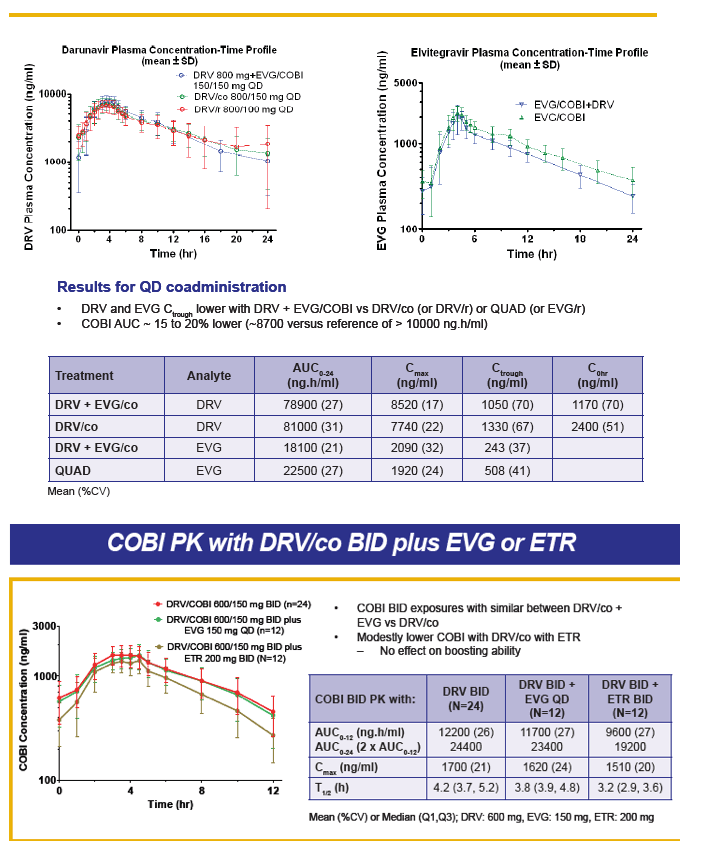
Safety/Tolerability, Pharmacokinetics, and Boosting of Twice-daily Cobicistat Administered Alone or in Combination with Darunavir or Tipranavir
|
| |
| |
Reported by Jules Levin
13th HIV Clinical Pharmacology Workshop Apr 16-18 2012 Barcelona Spain
S Ramanathan, H Wang, J Szwarcberg, and BP Kearney
Gilead Sciences, Inc., Foster City, CA, USA

Twice-Daily Cobicistat Effect Differs With Darunavir and Tipranavir - written by Mark Mascolini - (04/25/12)
Below Excerpted from:
Clinical Pharmacology at the 13th Workshop on Clinical Pharmacology of HIV Therapy - Courtney V. Fletcher, Pharm.D. - (05/14/12)
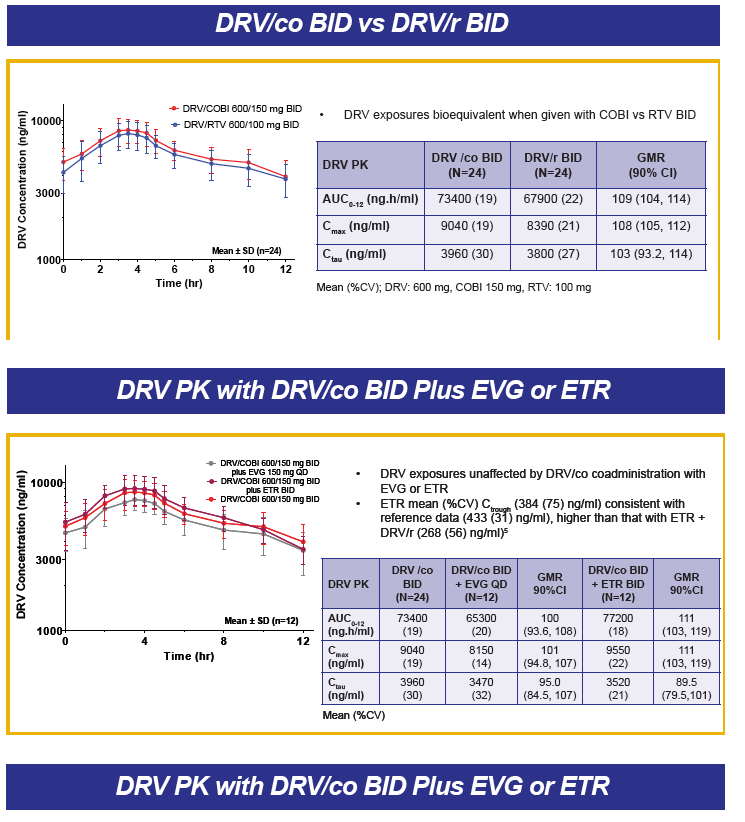
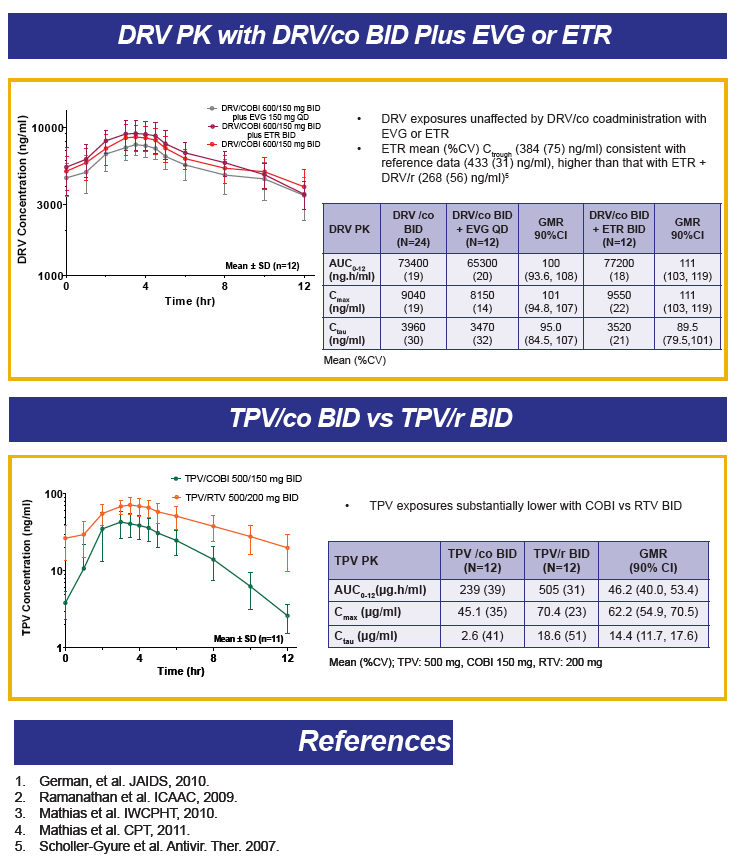
DRV PK in a fixed dose combination tablet with COBI are similar to those when given with RTV.
Abstract O_20 evaluated two different fixed dose combination tablet formulations of DRV with COBI in healthy volunteers (see also, http://www.natap.org/2012/pharm/Pharm_09.htm ). In the table below, I'll only provide data on the G004 formulation as that is the one selected for further development, although the PK of these two formulations appear bioequivalent.
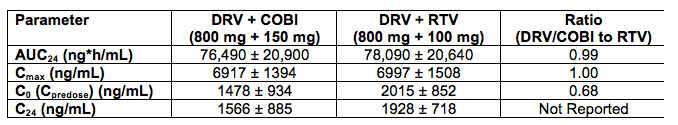
The AUC and Cmax of DRV when given with COBI or RTV are essentially the same. However, predose and 24 hour post dose concentrations, are approximately 32% lower on average with COBI than RTV. The abstract authors concluded this lower trough concentration was not clinically relevant. However, the study was done in healthy volunteers, in a fed state, in a rigorously controlled clinical environment. No data are available in HIV-infected persons, taking a different type of meal or no food at all, and with other common concomitant medications. Poster P_31 reported higher DRV trough concentrations when given once daily with RTV in the morning versus once daily with RTV in the evening (a circadian variation), indicating the time of drug administration is another variable that may affect concentrations. Jonathan Schapiro, M.D. made the point during the question and answer sessions that the burden of proof rests with the investigators to show these lower concentrations are truly not clinically relevant. I agree.
|
| |
|
 |
 |
|
|