 |
 |
 |
| |
Daily TDF/FTC PrEP Provides 86% Protection From HIV in Gay British Men
|
| |
| |
CROI 2015, February 23-26, 2015, Seattle, Washington
from jules: today at the opening session at CROI were 3 PrEP studies - 2 in MSM & a 3rd in couples in Africa reporting impressively good results, showing that PrEP works if adherence is good, as well an opening PrEP review by Rafael Landovitz, MD from LA that was very well done & I will report here his slide presentation. Here at CROI key subjects will be new HCV/HIV coinfection study results showing high SVR rates including ALLY-2 results which looked at daclatasvir+sofosbuvir across genotypes, the Harvoni study, Abbvie 3D data and simeprevir+sofosbuvir; heart disease in HIV+ will be a focus at this meeting too; and neurologic & cognitive impairment is a subject with much attention here as well.
Mark Mascolini
http://www.croiwebcasts.org/console/player/25539?mediaType=slideVideo&
Daily preexposure prophylaxis (PrEP) with tenofovir/emtricitabine (TDF/FTC) lowered HIV incidence 86% compared with deferred PrEP among gay/bisexual men in the open-label British PROUD trial [1]. That success rate mirrors protection from HIV in the French Ipergay trial, reported separately by NATAP, which deployed a 4-dose as-needed TDF/FTC strategy [2].
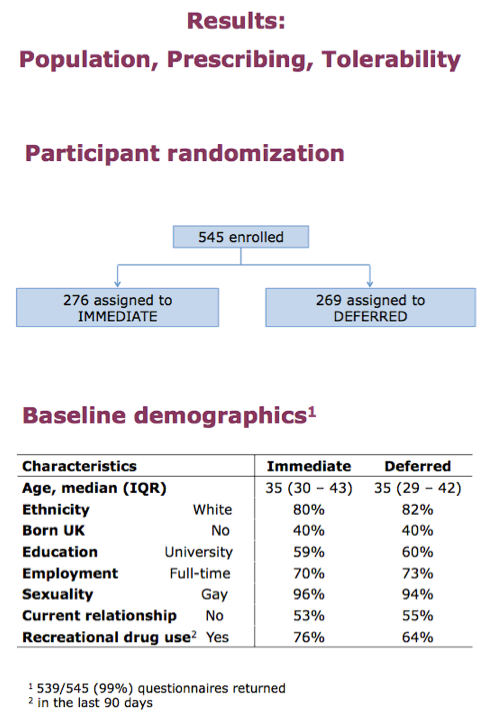
Because of concerns that PrEP success may encourage more frequent risky sex by PrEP users, PROUD investigators launched their trial to mimic real-world conditions among gay men using PrEP. Between November 2012 and April 2014 at 13 sexual health clinics in England, the researchers enrolled gay or bisexual men who reported condom-free anal intercourse in the past 90 days. The PROUD team randomized them to immediate open-label TDF/FTC or to defer starting TDF/FTC PrEP for 12 months. With follow-up every 3 months, the researchers planned a trial lasting up to 24 months.
Of the 545 men enrolled, 276 got assigned to immediate PrEP and 269 to deferred PrEP. Median age stood at 35 in both groups, about 80% in both groups were white, and 40% were not born in the UK. About 60% of participants had a university education, and nearly three quarters had a job. Slightly more than half of men said they had no current relationship. Majorities in both the immediate and deferred arms (76% and 64%) reported recreational drug use in the last 90 days.
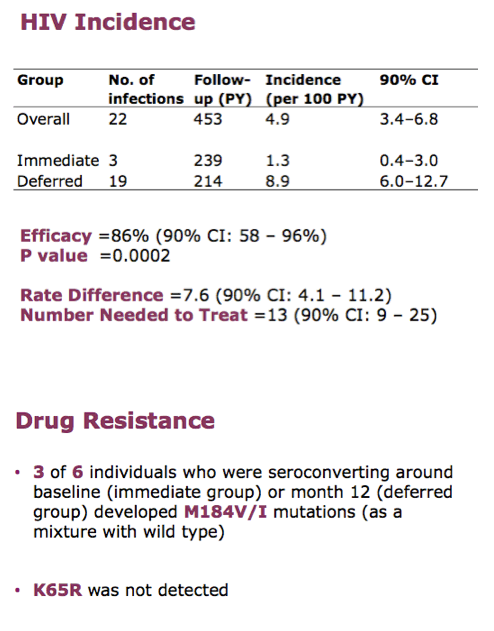
In October 2014 an independent review panel recommended offering men in the deferred arm immediate PrEP. Large majorities of men in both arms (97% immediate and 95% deferred) contributed to the effectiveness analysis. The PROUD team tallied 3 HIV infections in the immediate arm (2 in men who may have been seroconverting when the entered the trial) and 19 infections in the deferred arm. HIV incidence measured 1.3 per 100 person-years (90% confidence interval [CI] 0.4 to 3.0) with immediate PrEP and 8.9 per 100 person-years (90% CI 6.0 to 12.7) with deferred PrEP. Those rates translated into 86% efficacy with immediate PrEP (90% CI 58% to 96%, P = 0.002). Number needed to treat to prevent 1 infection in 1 year was 13.
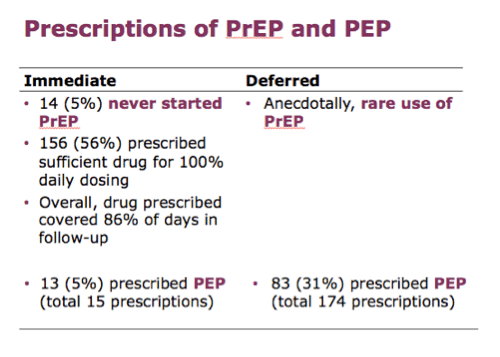
In the immediate arm, 5% of men never started PrEP. More than half in the immediate arm, 56%, got enough TDF/FTC prescribed for 100% daily dosing. Overall TDF/FTC prescriptions covered 86% of follow-up days. Pharmacokinetic sampling confirmed self-reports of good PrEP use.
During follow-up 5% in the immediate arm and 31% in the deferred arm got prescriptions for postexposure prophylaxis. Twenty-eight men overall interrupted PrEP, 13 of them for reasons considered related to study drug, including nausea and falling creatinine clearance. Eleven of those 13 restarted PrEP.
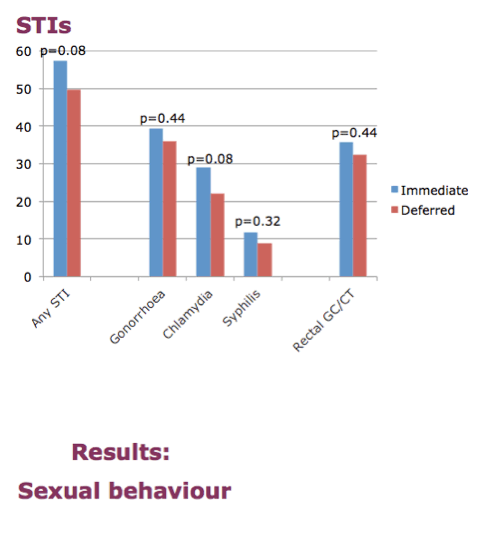
Nearly 60% of men in the immediate arm and almost 50% in the deferred arm had any STI, a nonsignificant trend (P = 0.08). Proportions of men with a confirmed STI indicating anal sex without condoms--rectal chlamydia or gonorrhea--were around 30% in both the immediate and deferred arms. The researchers cautioned, though, that more men in the immediate group got screened for STIs during the study period. Median total number of partners in the last 90 days stayed around the same at month 12 as before the trial in both study arms: 10. Numbers of condomless receptive and insertive anal partners were lower but also similar in the two arms: about 3.
Those high condom-free sex rates partly explain the lofty HIV incidence in the deferred PrEP arm. The PROUD team noted that HIV incidence in this PrEP-seeking gay population "was much higher than predicted based on all [gay/bisexual men] attending sexual health clinics." But PROUD's primary result convinced the researchers that their "concerns about PrEP being less effective in the real world [than in trials] were unfounded." They also noted that clinics readily adapted their routines to incorporate PrEP.
PROUD investigators summarize their findings and provide other trial information at http://www.proud.mrc.ac.uk/.
References
1. McCormack S, Dunn D, on behalf of the PROUD Study Group. Pragmatic Open-Label Randomised Trial of Preexposure Prophylaxis: the PROUD study. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 22LB.
2. Molina JM, Capitant C, Spire B, et al. On demand PrEP with oral TDF-FTC in MSM: results of the ANRS Ipergay trial. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 23.








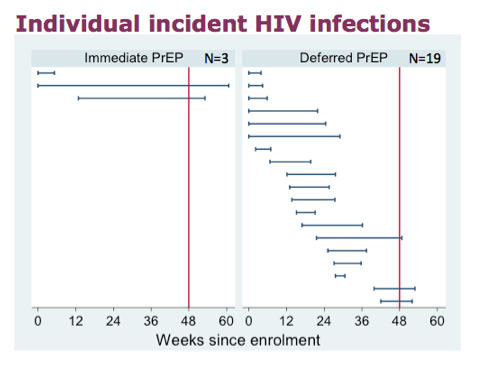
Individual incident infections with the most recent HIV negative result, and the first positive HIV result
The red bar indicates the time at which the window opened for the month 12 visit.
There were 2 infections in the immediate and 6 in the deferred diagnosed at the first follow-up visit, all of whom could have caught HIV before enrolment. If they were all dropped the effectiveness there would still be a huge difference between the groups.
Of the three immediate infections, one participant had a significant exposure in the week before enrolment which was too late for PEP. He took 15 doses of truvada before his HIV positive test. Although a sample taken 5 days later was negative for tenofovir in the plasma, this was as expected and the fact that he had the 184 minority mutation supports his story of taking Truvada.
The second individual did not return after enrolment and was diagnosed at a different clinic.
The third individual defaulted after three months and was later admitted to another hospital with a seroconversion illness. He told that hospital staff that he had not been taking PrEP for months and had no mutations.
86% reduction is greater than seen in placebo-controlled HIV prevention trials.
The 90% confidence interval gives us 95% confidence around the lower bound of 58% reduction. The 95% lower bound is 52% - both exceed the 50% reduction we considered would make a useful impact on our epidemic.
Rate difference is important for public health as it informs the number who would need to be treated. The number of gay men who need to be treated for one year to avert one infection is very low - only 13.

57% participants in the immediate group had an STI at some point in follow-up, and 50% of deferred participants.
35% immediate and 32% deferred had a rectal infection at least once during follow-up. NB the numbers in the abstract have been corrected.
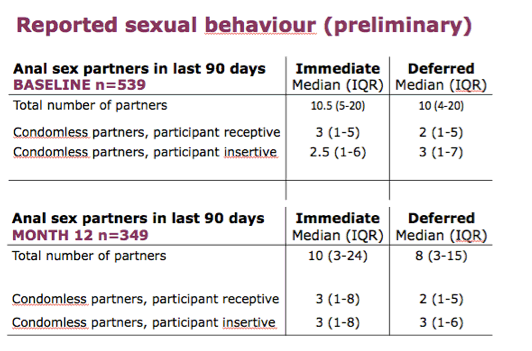
Ongoing analyses as we are still collecting month 12 questionnaires, but with this preliminary dataset the picture at month 12 is very similar to baseline for the median values.
The upper end of the distribution - the most sexually active - is important as it could be a key driver of the epidemic and there is some suggestion of a difference in the upper quartile between the groups, but no significant differences at this stage and we have to be very cautious about interpreting these results as the data are incomplete.
|
| |
|
 |
 |
|
|