 |
 |
 |
| |
Phase 1 Safety & PK Trial of Polyurethane Tenofovir Disoproxil Fumarate Vaginal Ring
|
| |
| |
Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
Marla J. Keller1, Pedro M. Mesquita1, Mark A. Marzinke2, Ryan Teller3, Bruce Frank4, Mark Mitchnick4, Peter L. Anderson5, Craig W. Hendrix2, Patrick F. Kiser3, and Betsy C. Herold1
1Albert Einstein College of Medicine, Bronx, New York; 2Johns Hopkins School of Medicine, Baltimore, MD; 3Northwestern University, Evanston, IL; 4Particle Sciences, Inc., Bethlehem, PA;
5University of Colorado Denver, Denver, Colorado
program abstract
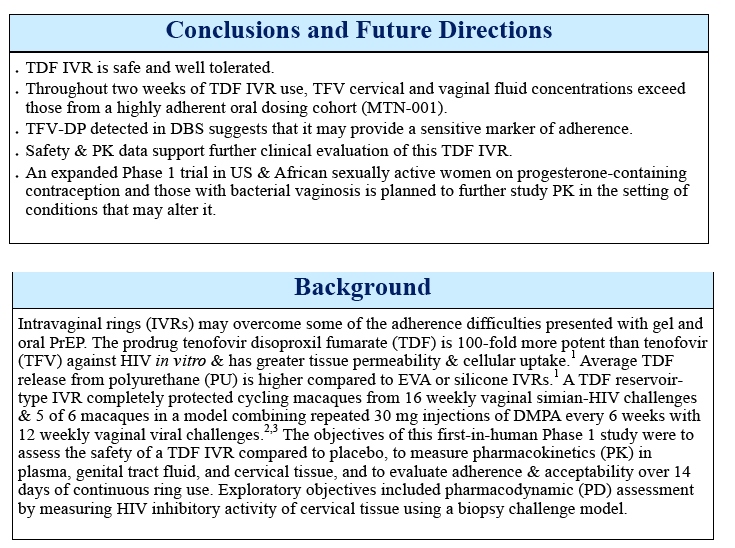
Background: The prodrug tenofovir disoproxil fumarate (TDF) is more potent than tenofovir (TFV) against HIV in vitro & has greater tissue permeability & cellular uptake. Sustained TDF delivery from a vaginal ring (VR) completely protected macaques from multiple vaginal simian-HIV challenges. The objectives of this trial were to evaluate safety & PK of reservoir-type TDF & placebo polyurethane VRs when used continuously for 14 days (d).
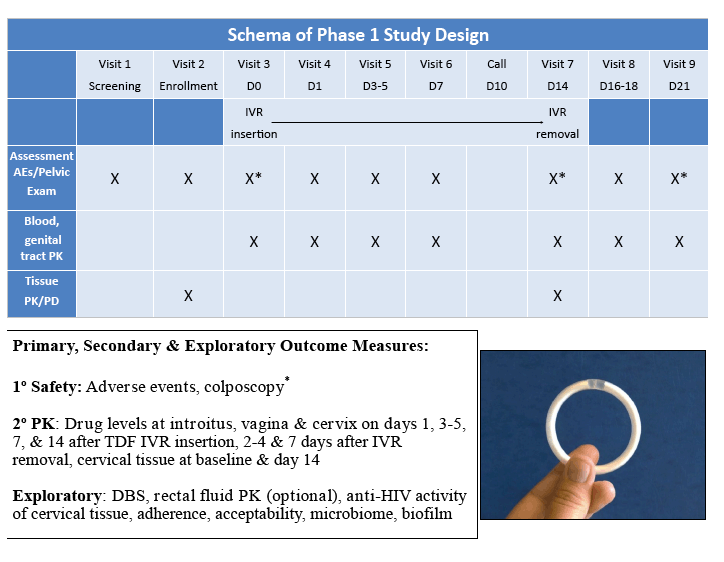
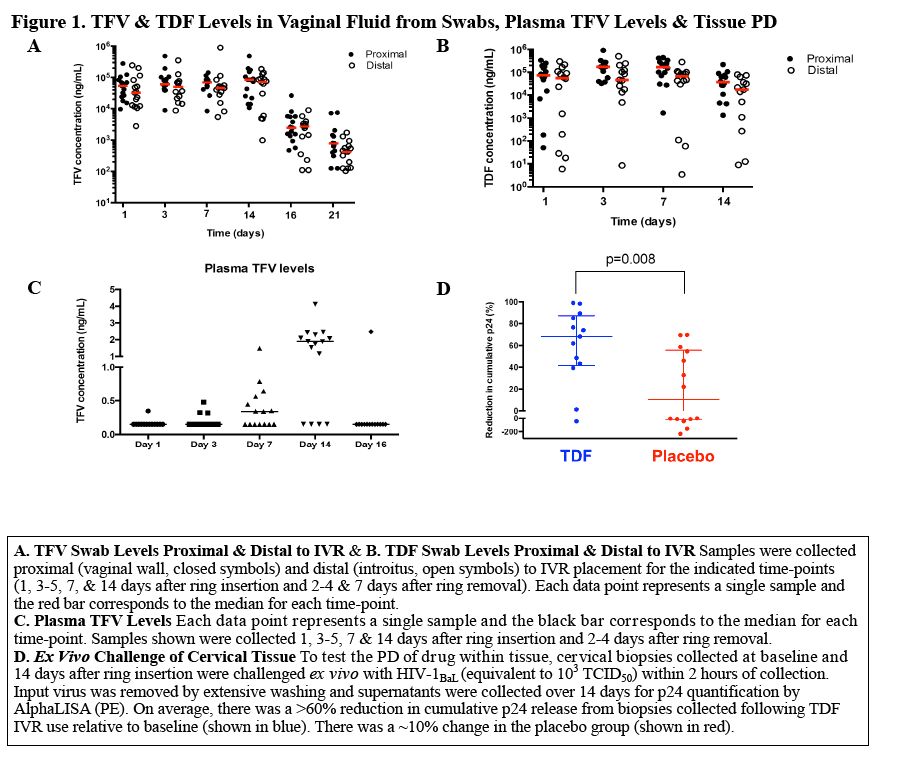
Methods: A randomized, single blind, placebo controlled trial of 30 women was conducted. Clinical safety was assessed. Swabs (2 proximal, 1 distal to VR) & plasma were collected 1, 3, 7 & 14 d after VR insertion & 2 & 7 d after removal for drug levels. Dried blood spots (DBS) were obtained after 7 & 14 d of VR use. Cervical tissue was collected for TFV, TFV-diphosphate (TFV-DP) & ex vivo HIV challenge studies. Tissue HIV LTR DNA in relative copy number was quantified by real-time PCR. To measure anti-HIV activity of luminal drug, Jurkat-Tat-CCR5 cells were challenged ex vivo with HIV-1BaL in the presence of cervical swab eluants. p24 levels in supernatants were assessed by AlphaLISA.
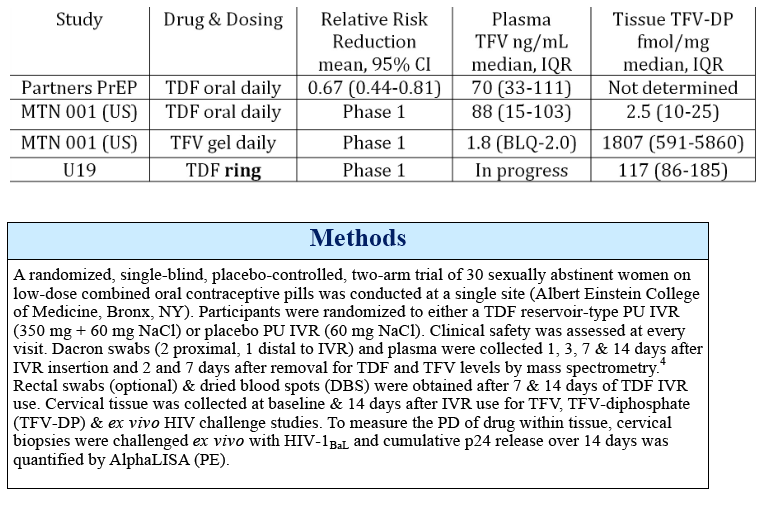
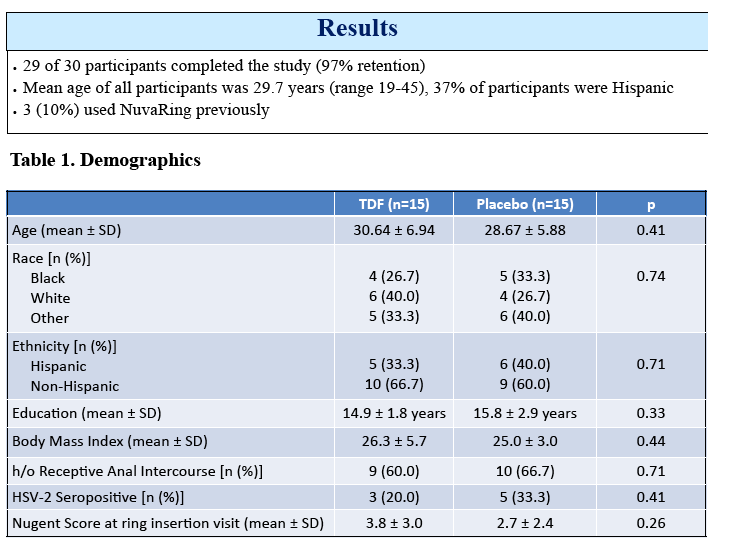
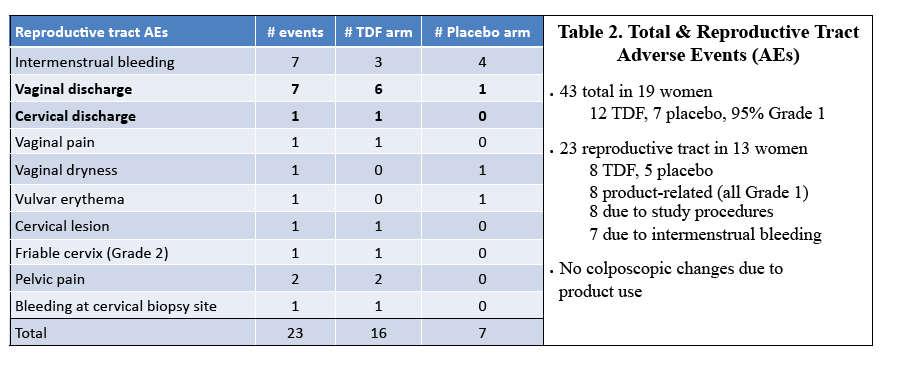
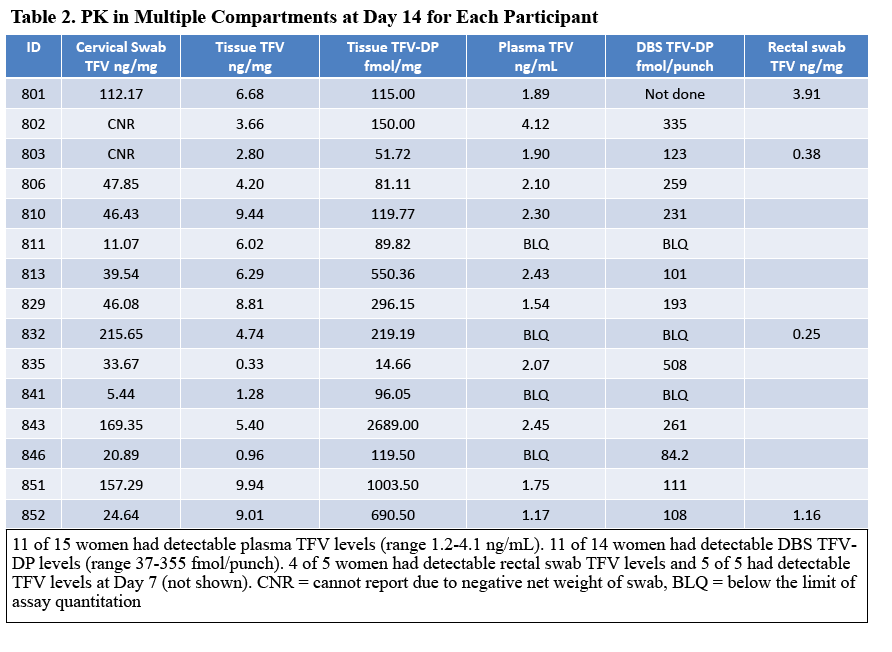
Results: 29 of 30 women completed the study (15 TDF, 14 placebo). The mean age was 29.7 years. There were 43 adverse events; 8 were product related & were Grade 1. Median TFV vaginal fluid (VF) levels in 10 women at 1, 3, 7 & 14 d after VR insertion were 4, 5, 7 & 7.3 x 104 ng/mL, respectively. Only 1 had detectable TFV-DP by DBS after 7 d but 11 of 14 had detectable levels after 14 d of TDF VR use (range 37-335 fmol/punch). Tissue TFV-DP levels in 10 women ranged from 52-550 fmol/mg. Tissue from women in TDF arm challenged ex vivo had a 90% reduction in HIV copy number compared to no change in the placebo arm (p
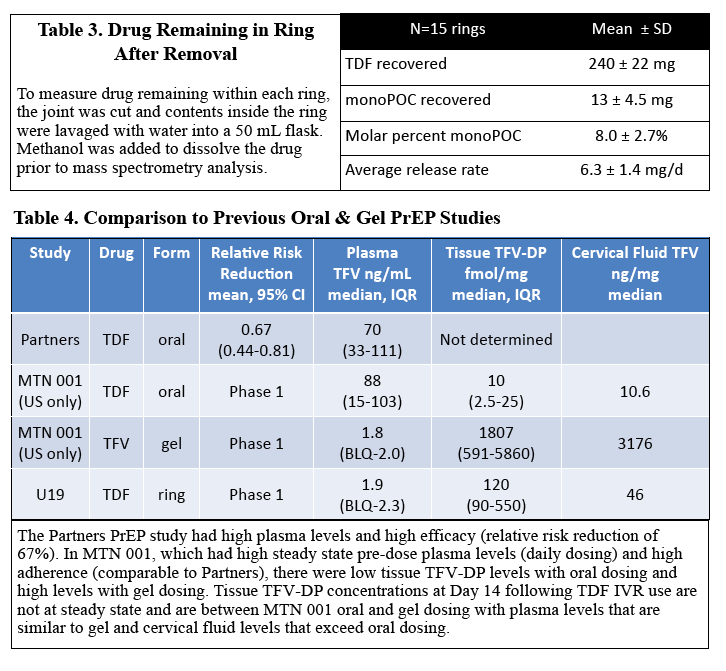
Conclusions: A TDF VR is safe, well tolerated & resulted in TFV VF levels that exceed the clinical correlate of protection observed with TFV gel BAT24 dosing. TFV-DP detected in DBS suggests that it may provide a sensitive maker of adherence. Findings support further development of this TDF VR.

|
| |
|
 |
 |
|
|