 |
 |
 |
| |
Rosuvastatin halts carotid artery wall thickening in placebo-controlled trial
|
| |
| |
CROI 2015, February 23-26, 2015, Seattle, Washington
Mark Mascolini
Webcast: http://www.croiwebcasts.org/console/player/25791?mediaType=slideVideo&
Daily rosuvastatin stopped progression of common carotid artery intima-media thickness (cIMT), a recognized predictor of atherosclerosis, in a double-blind placebo-controlled trial that enrolled adults with low-density lipoprotein (LDL) cholesterol below 130 mg/dL [1]. The impact of rosuvastatin on cIMT appeared to be independent of protease inhibitor use, insulin resistance, or changes in LDL cholesterol.
Grace McComsey and Case Western Reserve University colleagues who conducted this trial noted that the impact of statins on primary prevention of cardiovascular disease in people with HIV remains unknown. Some research indicates that statins can reduce or reverse cIMT progression in HIV-negative people, although these drugs appear to be more effective in secondary prevention.
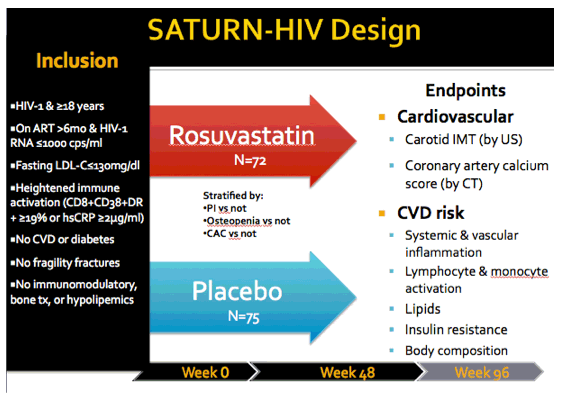
As part of the placebo-controlled SATURN-HIV trial, the Case Western team measured cIMT changes in the two study arms. SATURN enrolled HIV-positive adults taking antiretroviral therapy for more than 6 months. Everyone had a viral load at or below 1000 copies and fasting LDL ("bad") cholesterol at or below 130 mg/dL. Enrollees also had evidence of heightened immune activation. No one had cardiovascular disease, diabetes, or fragility fractures.
Previously reported effects of rosuvastatin in SATURN include decreased monocyte and lymphocyte activation, decreased NT-proBNP and Lp-PLA2 (predictors of cardiovascular disease in HIV-negative people), and increased insulin resistance [2-4]. The primary endpoint of the new analysis was cIMT change in the common carotid artery from baseline to week 96. Secondary endpoints were changes in carotid plaque and coronary artery calcification.
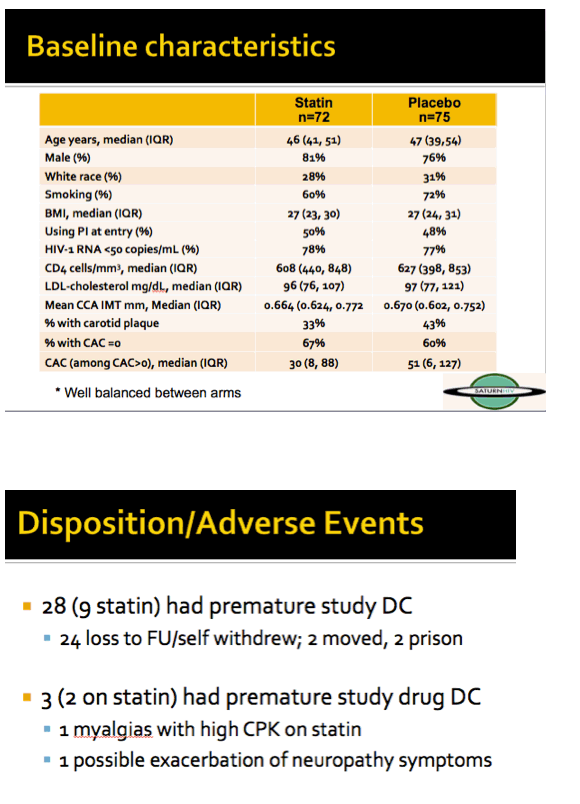
The 72 people randomized to rosuvastatin (10 mg daily) and the 75 randomized to placebo had median ages of 46 and 47. Men accounted for about 80% of the study group, and about 70% were black. Smokers made up 60% of the rosuvastatin group and 72% of the placebo group. Half of participants in each arm were taking a PI when they entered the study, and three quarters had a viral load below 50 copies. Median CD4 counts in the two study arms were above 600. Median LDL cholesterol measured 96 mg/dL in the statin group and 97 mg/dL in the placebo group. One third in the statin arm and 43% in the placebo arm had carotid plaque.
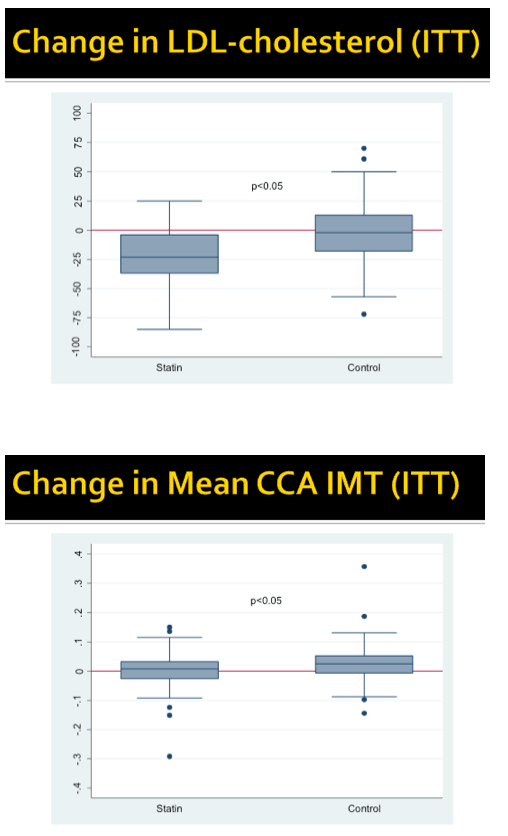
Two people randomized to rosuvastatin and 1 randomized to placebo stopped their study drug early. LDL cholesterol fell 21 mg/dL through 96 weeks in the statin arm while remaining nearly unchanged in the placebo arm, a significant difference (P < 0.05).
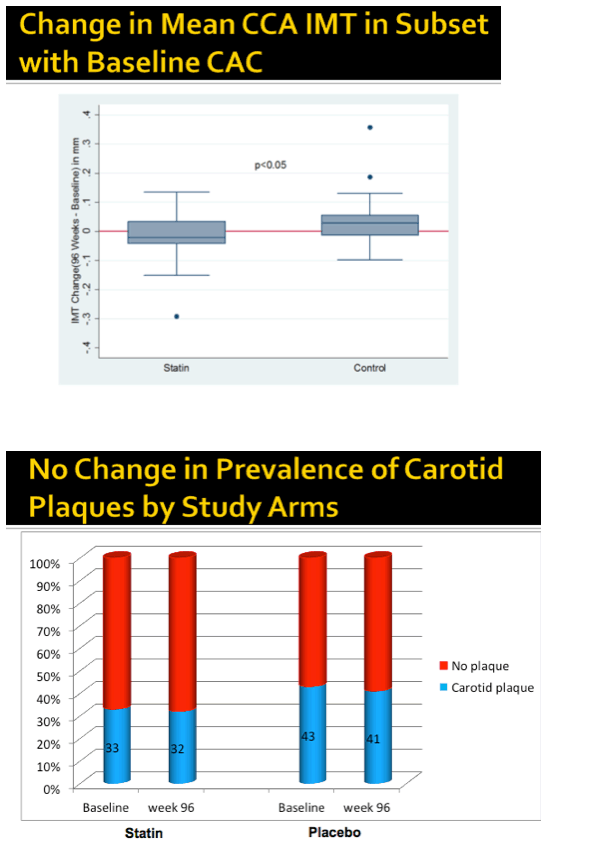
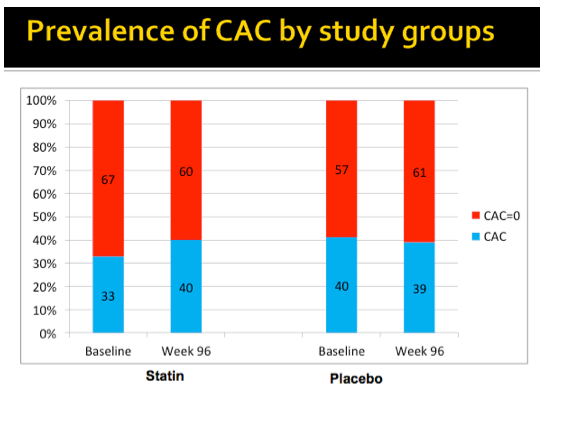
Average cIMT changed hardly at all through 96 weeks in the rosuvastatin group while climbing a fraction of a millimeter in the placebo arm, but the difference was statistically significant (P < 0.05). Prevalence of carotid plaque remained stable from baseline to week 96 in the statin group (33% and 32%) and in the placebo group (43% and 41%).
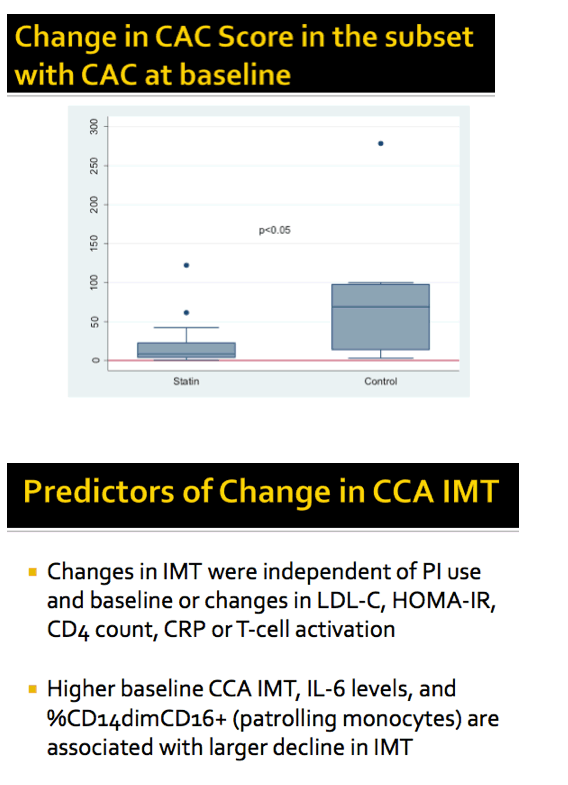
In people with baseline coronary artery calcification, cIMT dropped fractionally in the statin group and rose slightly in the placebo group, a significant difference (P < 0.05). In the subgroup with coronary artery calcium at baseline, coronary artery calcium score remained around the same through 96 weeks in the statin arm while rising substantially in the placebo arm (P < 0.05).
Three factors predicted a bigger drop in cIMT: higher baseline cIMT, levels of IL-6 (an inflammation marker), and percentage of patrolling monocytes (CD14dimCD16 cells). Factors not associated with cIMT change were PI use and baseline levels or change in LDL cholesterol, insulin resistance (by HOMA-IR), CD4 count, C-reactive protein (an inflammation marker), and T-cell activation.
McComsey and colleagues concluded that "rosuvastatin arrests IMT progression" in HIV-positive people on antiretroviral therapy with LDL cholesterol below 130 mg/dL. In people with coronary artery calcification at baseline, they added, "rosuvastatin halts the progression of coronary artery calcium."
Another placebo-controlled trial detailed at this CROI session and reported separately by NATAP found that 1 year of atorvastatin significantly cut coronary artery plaque volume and number in HIV-positive people with subclinical atherosclerosis [5].
References
1. Longenecker CT Jiang Y, Debanne SM, et al. Rosuvastatin arrests progression of carotid intima-media thickness in treated HIV. CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 137.
2. Funderburg NT, Jiang Y, Debanne SM, et al. Rosuvastatin treatment reduces markers of monocyte activation in HIV-infected subjects on antiretroviral therapy. Clin Infect Dis. 2014;58:588-595.
3. Funderburg NT, Jiang Y, Debanne SM, et al. Rosuvastatin reduces vascular inflammation and T-cell and monocyte activation in HIV-infected subjects on antiretroviral therapy. J Acquir Immune Defic Syndr. 2015;68:396-404.
4. Dirajlal-Fargo S, Kinley B, Jiang Y, et al. Statin therapy decreases N-terminal pro-B-type natriuretic peptide in HIV: randomized placebo-controlled trial. AIDS. 2015;29:313-321.
5. Lo J, Lu MT, Ihenachor EJ, et al. Effects of statin therapy on coronary artery plaque volume and high-risk plaque morphology in HIV-infected patients with subclinical atherosclerosis: a randomised, double-blind, placebo-controlled trial. Lancet HIV. 2015;2:e52-e63.



|
| |
|
 |
 |
|
|