 |
 |
 |
| |
Exposure to antiretrovirals and development
of chronic kidney disease - D:A:D Study Group
|
| |
| |
Tenofovir, LPV, ATV Linked to Higher CKD Rates Through 6 Years
CROI 2015, February 23-26, 2015, Seattle, Washington
webcast:http://www.croiwebcasts.org/console/player/25796?mediaType=slideVideo&
Mark Mascolini
Incidence of chronic kidney disease (CKD) continued to rise through 6 years of treatment with tenofovir, lopinavir/ritonavir, or atazanavir/ritonavir, according to a 23,560-person analysis of the D:A:D Study cohort [1]. People who had stopped tenofovir for 2 years still had a higher incidence of CKD than people who never started tenofovir.
Amanda Mocroft and D:A:D colleagues noted that several studies have linked tenofovir, ritonavir-boosted protease inhibitors (PIs), and abacavir to renal impairment. But it remains unclear whether the impairment represents only an "early hit" against the kidneys, they explained, or whether risk mounts with continuing use of these drugs.
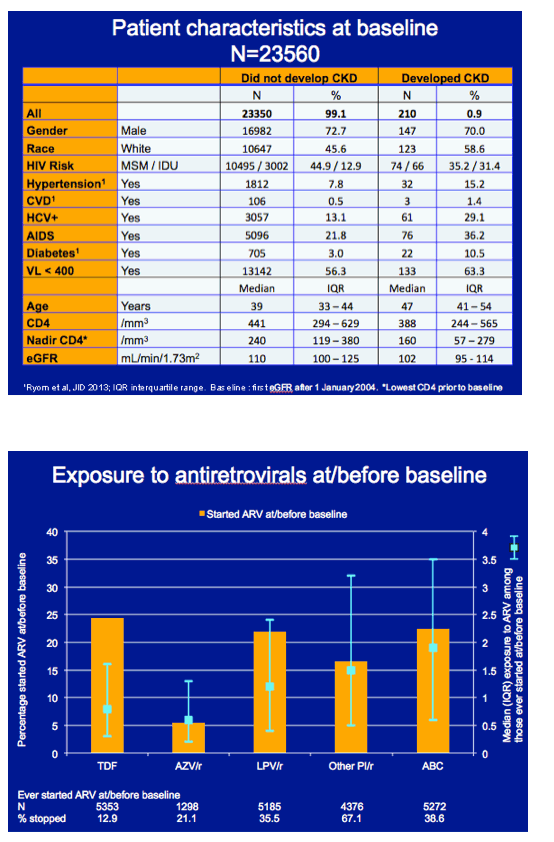
To address this issue, the D:A:D team assessed the association between tenofovir, atazanavir/ritonavir, lopinavir/ritonavir, other boosted PIs, and abacavir and CKD over time in people with initially normal kidney function indicated by an estimated glomerular filtration rate (eGFR) above 90 mL/min. The analysis included people with a baseline eGFR above that level after January 1, 2004.
Follow-up continued until CKD (confirmed eGFRs below 60 mL/min more than 3 months apart), their last eGFR, their last visit plus 6 months, or January 1, 2013. The researchers excluded people with fewer than two eGFRs after the baseline measure. Mocroft and colleagues used Poisson regression adjusted for age, gender, race, and many other variables to estimate CKD associated with (1) cumulative exposure to the antiretrovirals of interest or (2) time since stopping those drugs.
The analysis included 23,560 cohort members, 210 (0.9%) of whom had CKD develop during a median 6.3 years of follow-up. Men made up about 70% of the groups with and without CKD, while whites made up 59% of the CKD group and 46% of the no-CKD group. HCV prevalence was 29% in the CKD group and 13% in others. Respective diabetes rates were 10.5% and 3%. Median age measured 47 in people with CKD and 39 in those without CKD. Median nadir CD4 count stood at 160 in the CKD group and 240 in the other group.
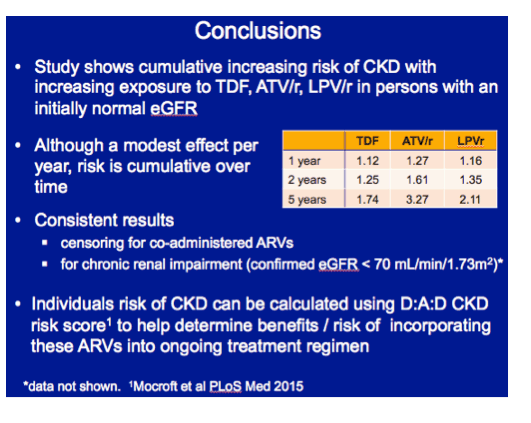
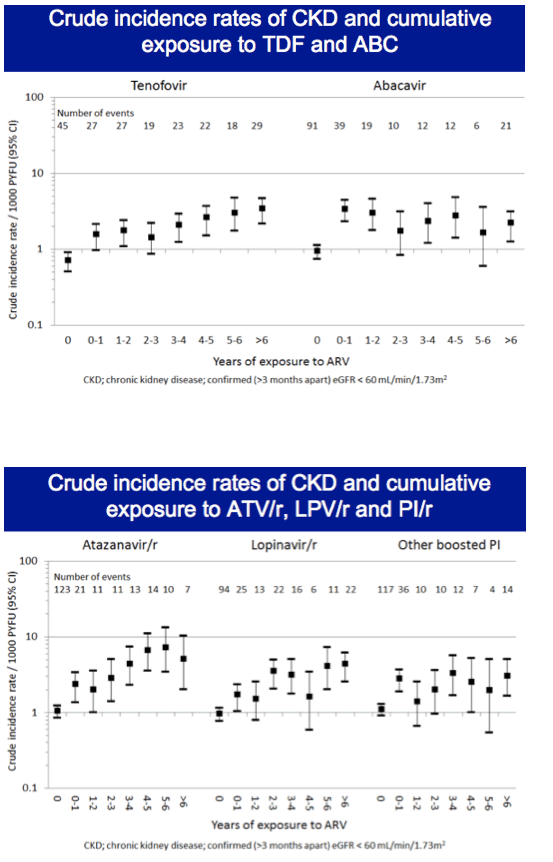
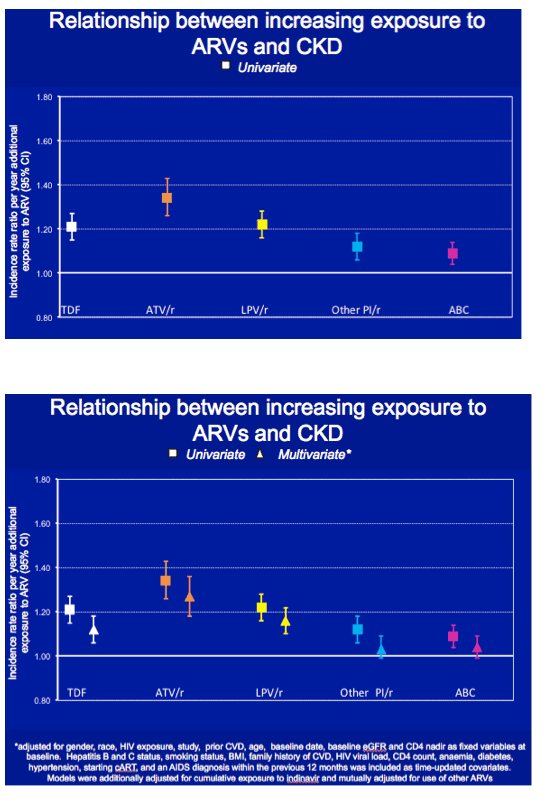
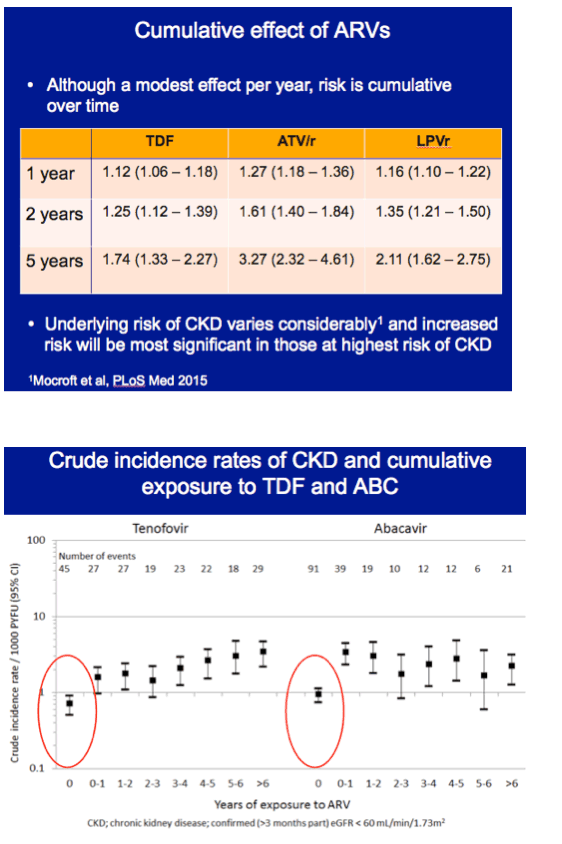
In multivariate analysis, each additional year of tenofovir exposure was associated with a 12% higher CKD incidence (adjusted incidence rate ratio [aIRR] 1.12, 95% confidence interval [CI] 1.06 to 1.18), Each year of atazanavir/ritonavir was linked to almost a 30% higher CKD incidence (aIRR 1.27, 95% CI 1.18 to 1.36), and every added year of lopinavir/ritonavir translated into a 16% higher CKD incidence (aIRR 1.16, 95% CI 1.10 to 122). Multivariate analysis did not tie cumulative use of other boosted PIs or abacavir to higher CKD incidence.
With tenofovir, atazanavir/ritonavir, and lopinavir/ritonavir, CKD incidence did not level off after the first year of use. Respective adjusted incidence rate ratios were 1.25, 1.61, and 1.35 at 2 years and 1.74, 3.27, and 2.11 at 5 years.
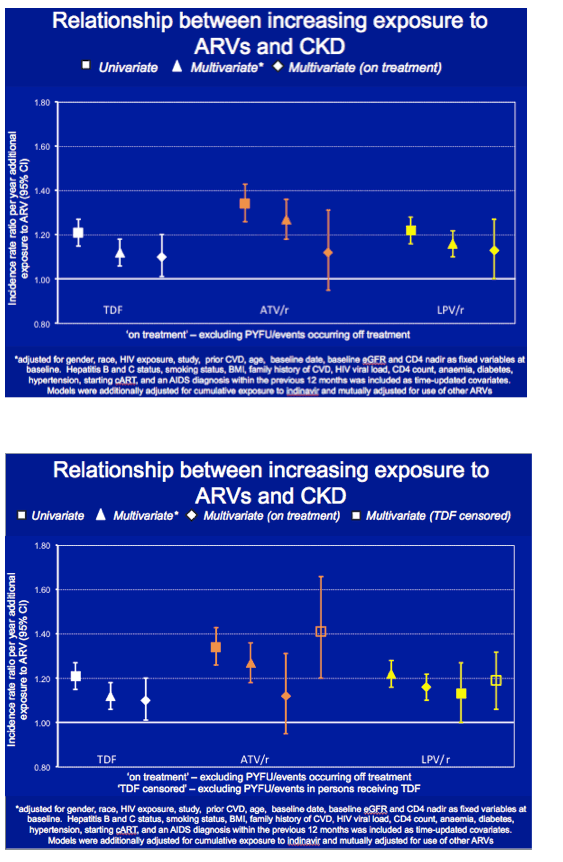
When the D:A:D investigators limited the analyses of tenofovir, lopinavir, and atazanavir to people currently on treatment, the adjusted incidence rate ratio per year of treatment declined from the initial adjusted result for atazanavir and crossed 1.0 but hardly changed for tenofovir or lopinavir. Eliminating tenofovir from atazanavir and lopinavir combinations hardly changed the lopinavir adjusted incidence rate ratio, while the incidence rate ratio for each year of atazanavir climbed to about 1.4.
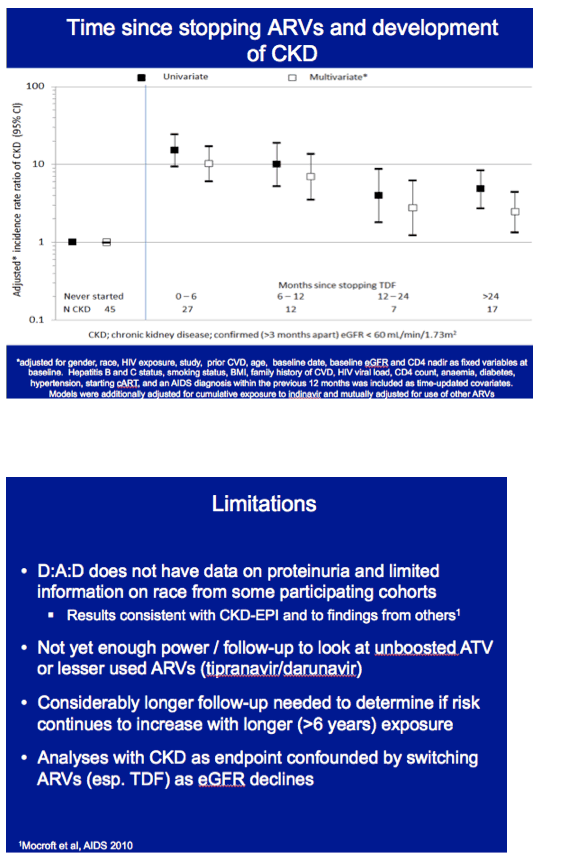
CKD adjusted incidence rate ratios fell steadily with time since stopping tenofovir. But after tenofovir had stopped more than 24 months, the 95% confidence interval around the adjusted incidence rate ratio for CKD still did not descend below 1.0. In contrast, among people who never started tenofovir, the adjusted incidence rate ratio for CKD stood squarely at 1.0.
These analyses could not consider unboosted atazanavir or darunavir/ritonavir because of limited statistical power or insufficient follow-up.
The researchers concluded that 1 year of treatment with tenofovir, lopinavir/ritonavir, or atazanavir/ritonavir had only a modest impact on CKD incidence, but that incidence continued to grow through 5 years in adjusted analysis. Results with these three antiretrovirals remained consistent when the analysis focused on chronic renal impairment, defined as a confirmed eGFR below 70 mL/min.
Reference
1. Mocroft A, Lundgren JD, Ross M, et al. Exposure to antiretrovirals (ARVs) and development of chronic kidney disease (CKD). CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 142.
---------------------------------
Exposure to antiretrovirals and development of chronic kidney disease
Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
Amanda Mocroft, Jens Lundgren, Michael Ross, Christoph Fux, Peter Reiss, Olivier Moranne, Philippe Morlat, Antonella d'arminio Monforte, Ole Kirk, Lene Ryom on behalf of the D:A:D* study group




|
| |
|
 |
 |
|
|