 |
 |
 |
| |
SVR Durability: HCV Patients Treated with IFN-Free DAA Regimens
|
| |
| |
These data reflect 6 studies with sofosbuvir/Rbv or Harvoni conducted by the DC based NIH group, 85% of study participants are African-Americans, 69% coinfected, 31% monionfected, 82% CT/TT, 25% fibrosis 3/4 HAI Score.
Reported by Jules Levin
CROI 2015 Feb 23-26, Seattle, WA
Aurielle Thomas1, Sarah Kattakuzhy2, Sara Jones3, Anita Kohli4, Eleanor Wilson2, Angie Price2, Rachel Silk2, Zayani Sims4, Anu Osinusi5, Shyam Kottilil2
1 Laboratory of Immunoregulation, National Institute of Allergy and Infectious Diseases, National Institutes of Health, 2Division of Infectious Diseases, Institute of Human Virology, University of Maryland, 3Clinical Research Directorate/Clinical Monitoring Research Program, Leidos Biomedical Research, Inc., 4Critical Care Medicine Department, National Institutes of Health Clinical Center, Frederick National Laboratory for Cancer Research, 5Gilead Sciences, Inc., Foster City, CA,


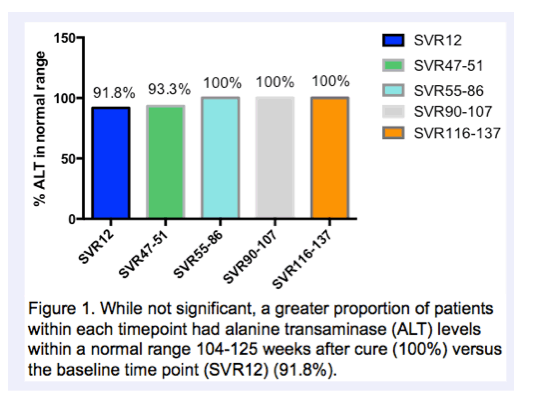

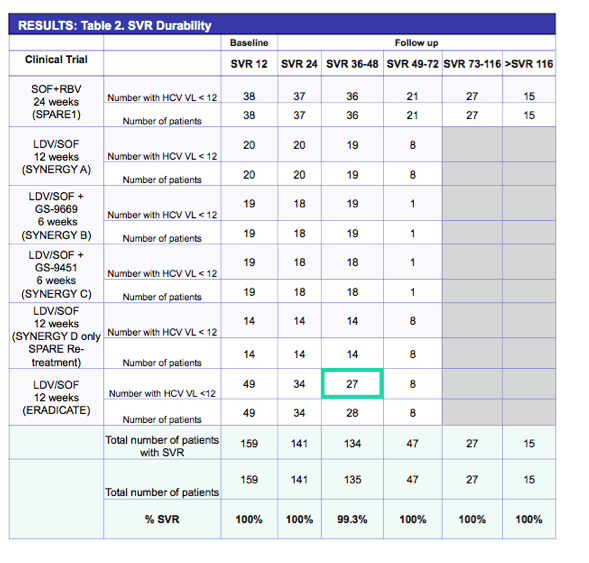
|
| |
|
 |
 |
|
|