 |
 |
 |
| |
CXCR4-using HIV--a tiny population before
stem-cell transplant-becomes major population after
|
| |
| |
Download the PDF
Full poster below and published NEJM Letter from author attached
CROI 2015, February 23-26, 2015, Seattle, Washington
Mark Mascolini
A CXCR4-using HIV variant detected as a minor population in a lymphoma patient before allogeneic hematopoietic stem-cell transplant (HSCT) emerged as the major population after the transplant with stem cells from a donor homozygous for the CCR5 delta32 mutation [1]. Researchers from the University of Duisburg-Essen believe selective pressure exerted by the transplant explains the emergence of the minor CXCR4 variant.
The only verified cure of HIV infection followed myeloablation and subsequent allogeneic HSCT with cells from a donor homozygous for (carrying two copies of) the delta32 mutation in CCR5 [2]. That mutation makes cells invulnerable to infection with CCR5-using virus, but people carrying the CCR5 mutation remain vulnerable to CXCR4-using HIV. Since that report of the Berlin patient, who had leukemia, at least 6 more HIV-positive men with lymphoma or leukemia have had allogenic HSCT with cells from a donor homozygous for the rare delta32 mutation, according Gero Hutter, who led the Berlin team [3].
All of these 6 men died within a year of their stem-cell transplant, including a man treated at the University of Duisburg-Essen by Jens Verheyen and colleagues. The Duisburg-Essen team reported earlier that CXCX4-using HIV emerged as the major HIV population after this transplant [4]. At CROI Verheyen and coworkers offered a detailed analysis of HIV RNA and DNA sequencing in samples collected before and after myeloablation and HSCT with delta32 stem cells [1].
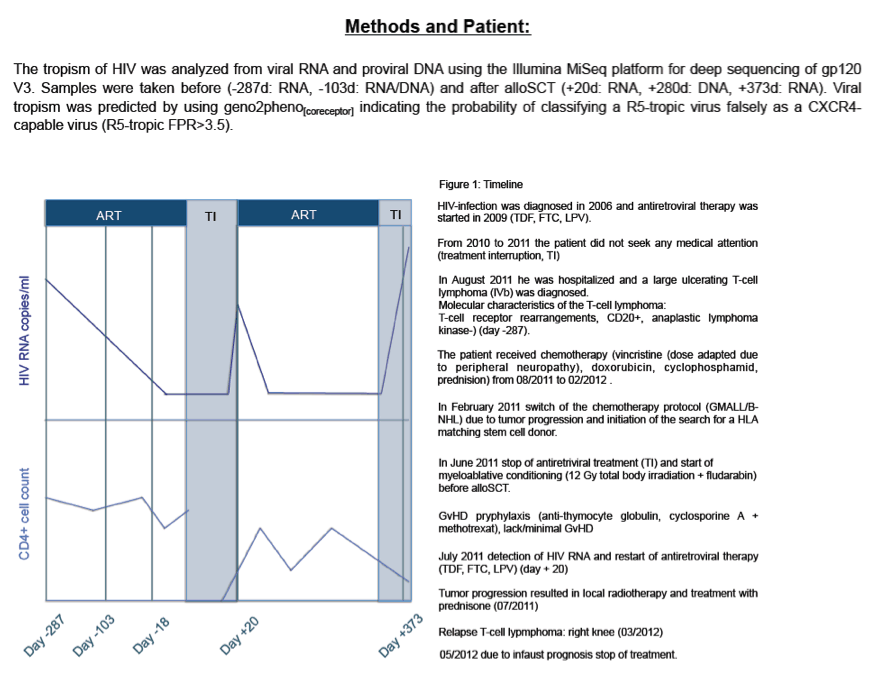
Clinicians diagnosed HIV in this man in 2006. He started antiretroviral therapy in 2009 but dropped out of care and received no treatment for a period from 2010 to 2011. The man entered a hospital in 2011 with a large ulcerating T-cell lymphoma. Two course of chemotherapy failed to arrest tumor development. In June 2011 he stopped antiretroviral therapy and began myeloablative conditioning including total body irradiation and fludarabine, then had allogeneic HSCT. With detectable HIV RNA in July 2011, the man resumed antiretroviral therapy. The tumor continued to progress, antiretroviral therapy stopped, and the man died.
In a previous publication on the case, Verheyen and coworkers reported that CXCR4-using HIV had emerged upon post-transplant rebound in their patient. This shift from a mainly CCR5-using HIV population before HSCT to a mainly CXCR4-using population afterwards, they wrote, "was probably driven by transplantation with stem cells homozygous for the CCR5 delta32 mutation" [4]. The Duisberg-Essen team suggested their case "highlights the fact that viral escape mechanisms might jeopardize CCR5-knockout strategies to control HIV infection" [4].
To explore these findings further, the Duisberg-Essen investigators determined HIV tropism (preference for CXCR4- or CCR5-using cells) in viral RNA and/or proviral DNA with a deep-sequencing technique in samples collected 287 and 103 days before HSCT and 20, 280, and 373 days after HSCT. They predicted viral tropism with the geno2pheno(coreceptor) tool.
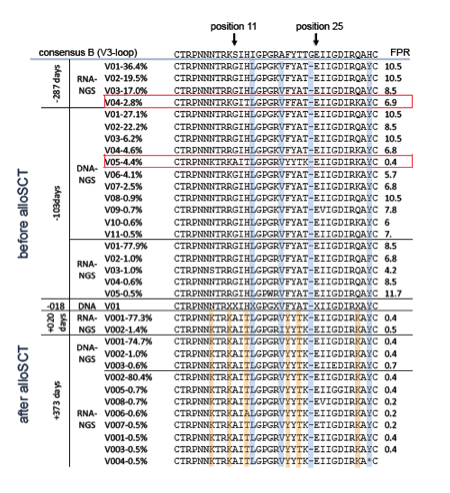
One HIV variant detected as 4.4% of the HIV DNA population before the transplant proved identical to the dominant HIV variant replicating in the patient after HSCT. The geno2pheno tool classified this variant as "clearly X4-capable."
Verheyen and colleagues concluded that "even the presence of minor X4-capable variants can be responsible for lack of control of HIV replication from therapy regimens aiming at the functional knock-out of the cellular CCR5 coreceptor of immune cells" [1].
In a published exchange of letters before the CROI report, Gero Hutter suggested that CXCR4-using virus may not have emerged if the Essen patient remained on suppressive antiretroviral therapy [3]. "A lesson to be taken from the [Essen] case," he proposed, "may be to continue the administration of antiretroviral therapy during transplantation until a stable donor chimerism is achieved" [3].
References
1. Verheyen J, Dirks M, Widera M, et al. Breakthrough of preexisting X4-capable HIV after allogeneic stem-cell transplantation CROI 2015. February 23-26, 2015. Seattle, Washington. Abstract 431.
2. Hutter G, Nowak D, Mossner M, et al. Long-term control of HIV by CCR5 delta32/delta32 stem-cell transplantation. N Engl J Med. 2009;360:692-698. http://www.nejm.org/doi/full/10.1056/NEJMoa0802905
3. Hutter G. More on shift of HIV tropism in stem-cell transplantation with CCR5 delta32/delta32 mutation. N Engl J Med. 2014;371:2438. http://www.nejm.org/doi/full/10.1056/NEJMc1412279
4. Kordelas L, Verheyen J, Beelen DW, et al. Shift of HIV tropism in stem-cell transplantation with CCR5 delta32 mutation. N Engl J Med. 2014;371:880-882. http://www.nejm.org/doi/full/10.1056/NEJMc1405805
-----------------------------
NEJM published Letter by authors below
Reported by Jules Levin
Breakthrough of Preexisting X4-capable HIV After Allogeneic Stem-Cell Transplantation
Verheyen Jens (1), Thielen Alexander (2), Sichtig Nadine (3), Dirks Miriam (1), Widera Marek (1), Kordelas Lambros (4), Daumer Martin (2), Kaiser Rolf (3), Esser Stefan (5)
1) Institute of Virology, University Hospital, University of Duisburg-Essen, Essen, Germany, 2) Institute of Immunology and Genetics, Kaiserslautern, Germany, 3) Institute of Virology, University of Cologne, Cologne, Germany, 4) Department of Bone Marrow
Transplantation, University Hospital, University of Duisburg-Essen, Essen, Germany, 5) Clinic for Dermatology, University Hospital, University of Duisburg-Essen, Essen, Germany

program abstract
Breakthrough of Preexisting X4-Capable HIV After Allogeneic Stem-Cell Transplantation
Primary Author:
Jens Verheyen1, Miriam Dirks1, Marek Widera1, Lambros Kordelas1, Stefan Esser1, Alexander Thielen2, Martin Daumer2, Nadine Lubke3, Rolf Kaiser3
1 Institute of Virology, University Hospital Essen, University Duisburg-Essen, Essen, Germany;2 Institute of Immunology and Genetics, Kaiserslautern, Germany;3 Institute of Virology, University of Cologne, Cologne, Germany
Abstract Body:
Background: Recently, we reported the case of an HIV-infected patient diagnosed with T-cell lymphoma who subsequently underwent allogeneic stem-cell transplantation (alloSCT) from a CCR5 delta32 homozygously mutated donor. In this case HIV replication could not be controlled by the immune system and the patient died after a relapse of the T-cell lymphoma. So far, the evolutionary pathways of the HIV tropism in this patient has remained unclear.
Methods: The tropism of HIV was analyzed from viral RNA and proviral DNA using the Illumina MiSeq platform for deep sequencing of gp120 V3. Samples were taken before (-287d: RNA, -103d: RNA/DNA) and after alloSCT (+20d: RNA, +280d: DNA, +373d: RNA). Viral tropism was predicted by using geno2pheno[coreceptor] indicating the probability of classifying a R5-tropic virus falsely as a CXCR4-capable virus (FPR).
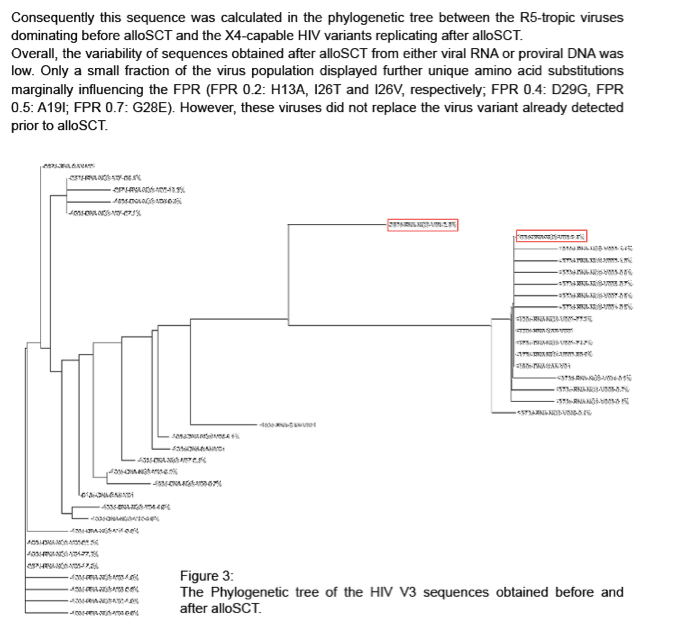
Results: Several distinct virus populations could be observed before and after alloSCT, which harbored specific mutational patterns (I14L, A19V, G24-, H34Y). Before alloSCT two virus populations were dominantly found in viral RNA and DNA only distinguished by mutation (R18K) with FPR of 10.5 (R18wt) and 8.5 (R18K), respectively. Apart from these two virus populations several minority variants could be detected carrying additional amino acid substitutions (N7S, K10R, H13T, R18W, Q32K, H34F) resulting in FPR from 7.8 to 4.2. These minority variants were especially detected in proviral DNA (-103d). One HIV variant detected in proviral DNA (4.4%) before alloSCT (-103d) had a unique mutational pattern (S11A, H13T, I14L, A19V, F20Y, T21K, Q32K) classified as clearly X4-capable (FPR 0.4). This sequence was identical to the sequence of the dominant HIV variant replicating in the patient after alloSCT. Overall, the viral variability was limited in sequences obtained after alloSCT from viral RNA and proviral DNA. Only a small fraction of the virus population displayed further unique amino acid substitutions marginally influencing the FPR (FPR 0.2: H13A, I26T and I26V, respectively; FPR 0.5: A19I; FPR 0.7: G28E).
Conclusions: The selective pressure exerted by the transplantation of allogeneic stem-cells homozygous for the CCR5 delta32 mutation resulted in the selection of already preexisting X4 capable HIV. Even the presence of only a minor X4 variant before alloSCT prevented the control of HIV in the absence functional CCR5 receptor.


------------------------------
Correspondence
Shift of HIV Tropism in Stem-Cell Transplantation with CCR5 Delta32 Mutation
N Engl J Med Aug 28 2014; 371:880-882
To the Editor:
Infection with the human immunodeficiency virus (HIV) requires entry into target cells by binding of the viral envelope to the CD4 receptor and to either the chemokine (C-C motif) receptor 5 (CCR5) or the chemokine (C-X-C motif) receptor 4 (CXCR4). Homozygosity for a 32-bp deletion in the CCR5 allele (CCR5 delta32) prevents cellular entry of CCR5-tropic (R5-tropic) HIV type 1 (HIV-1) strains. In 2009, there was a report1 about an HIV-1-infected patient with acute myeloid leukemia in whom the viral load remained undetectable after allogeneic stem-cell transplantation from a donor who was homozygous for the CCR5 delta32 mutation and after the discontinuation of antiretroviral therapy. This case gave rise to hope for new strategies for eradicating HIV-1 infection. However, this case has remained unique. Furthermore, in HIV-1-infected patients undergoing allogeneic stem-cell transplantation from donors with nonmutated CCR5, viral rebound has been reported.2
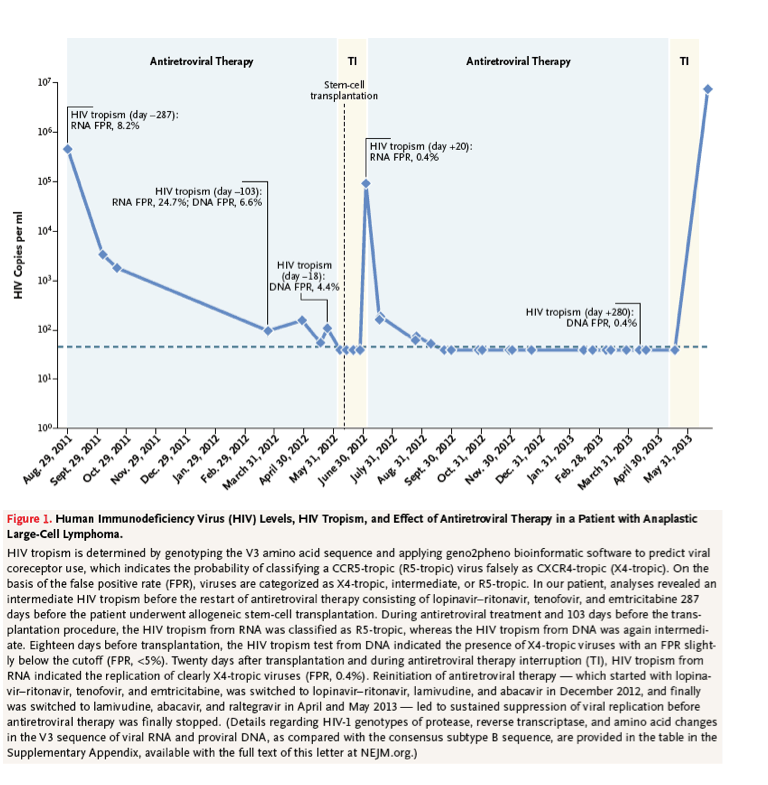
Here, we present the case of a 27-year-old patient with HIV-1 infection and anaplastic large-cell lymphoma. Because of a poor prognosis after progression of the T-cell lymphoma, stem-cell transplantation was planned, and a donor who was homozygous for the CCR5 delta32 mutation was identified. We determined the viral tropism of HIV-1 by genotyping the V3 amino acid sequence and applying geno2pheno bioinformatic software to predict viral coreceptor use,3 which indicates the probability of classifying an R5-tropic virus falsely as a CXCR4-tropic (X4-tropic) virus (false positive rate of <5%, X4-tropic; false positive rate of 5 to 10%, intermediate; false positive rate of >15%, R5-tropic). Before the patient underwent transplantation, the tropism from viral RNA was predicted to be either R5-tropic (false positive rate, 24.7%) or intermediate (false positive rate, 8.2%), whereas the V3 sequence from proviral DNA was classified as intermediate (false positive rate, 6.6%) or X4-tropic (false positive rate, 4.4%).
The patient discontinued antiretroviral therapy before the initiation of myeloablative treatment but resumed therapy 3 weeks after transplantation because of a rebound of 93,390 copies of HIV RNA per milliliter (Figure 1). The V3 sequence was related to the previous genotypes from this patient, as indicated by the presence of identical mutations in all V3 sequences (see the table in the Supplementary Appendix); it also carried several specific mutations resulting in the prediction of an X4-tropic virus (false positive rate, 0.4%). Antiretroviral therapy effectively suppressed viral replication until the patient had a relapse of the T-cell lymphoma, when antiretroviral therapy was again stopped. Two weeks before the patient died, the HIV-1 RNA level was 7,582,496 copies per milliliter.
The genotypic analyses of HIV-1 variants in this patient showed a shift from a dominantly R5-tropic HIV before stem-cell transplantation toward an X4-tropic HIV after transplantation. This shift of tropism was probably driven by transplantation with stem cells homozygous for the CCR5 delta32 mutation. This case highlights the fact that viral escape mechanisms might jeopardize CCR5-knockout strategies to control HIV infection.4
|
| |
|
 |
 |
|
|