| |
Effectiveness of DAA-based treatment of HCV in active people who inject
drugs living in middle income countries (MIC): the results of a prospective cohort study in Tbilisi, Georgia
|
| |
| |
EASL: The PREVAIL Study: Intensive Models of HCV Care for People Who Inject Drugs - (04/26/17)
EASL: Efficacy and safety of sofosbuvir/velpatasvir in people with chronic hepatitis C virus infection and recent injecting drug use: The SIMPLIFY study - (04/24/17)
HCV Treatment for IDUs and Comorbidities (Cancer, CVD, Kidney, Psych Issues) - (06/09/17)
APASL: C-EDGE CO-STAR: Interim Results From the 3-year Follow-up Trial on Risk Factors and Rate of Reinfection in Patients on Opiate Agonist Therapy Previously Treated With Elbasvir/Grazoprevir for 12 Week - (03/15/17)
AASLD: ELEVATED HCV REINFECTION INCIDENCE AFTER SUCCESSFUL TREATMENT AMONG HIV-INFECTED MEN WHO HAVE SEX WITH MEN IN SAN DIEGO - (11/21/16)
AASLD: Impact of Drug Use and Opioid Substitution Therapy on Hepatitis C Reinfection: The BC Hepatitis Testers Cohort - (11/21/16)
CROI/2016: Understanding the Relative Contributions of IDU and HCV on Systemic Immune Activation HCV should be aggressively treated in current IDUs even more if HIV and/or HCV positive - (04/4/16)
CROI: Peripheral endothelial function is suboptimal in HCV-untreated, HIV-suppressed adults - - (05/09/17)
Reported by Jules Levin
EASL Apr 20, 2017
Julie Bouscaillou, Tamar Kikvidze, Maïa Butsashvili, Konstantine Labartkava, Ina Inaridze, Aurelie Etienne,George Kamkamidze, Ani Gamezardashvili, Elisabeth Avril , Niklas Luhmann

Background and aims
Although they carry a high HCV burden globally, active people who inject drugs (PWID) are often excluded from national policies due to concerns about their ability to adhere to care, especially in MIC. Georgia faces high HCV rates (7.1% of antibodies in general population) with 25.6% of the cases being among PWID. An ambitious National HCV elimination Plan was launched in 2015, with initially 7000 treatments dedicated to patients with advanced liver fibrosis (≥F3). We assessed the treatment outcomes in PWID treated in the framework of the National Plan.
Methods
We followed a prospective cohort of PWID clients of a needle and syringe exchange program and supported by peer workers during treatment. PWID were treated with sofosbuvir and ribavirin +/-pegInterferon according to the genotype, treatment experience and level of fibrosis. We collected data concerning bio-medical parameters, adherence to care, demographics, and behaviors before and during treatment. After a descriptive analysis, we studied the factors associated with adherence to care and sustained virologic response at 12 weeks post-treatment (SVR12) using adjusted logistic regressions. We additionally compared the SVR12 rate to those of patients not reporting any history of injecting drug use (non-PWID), treated at the same clinic, during the same period - adjusting for age, sex, genotype, level of fibrosis, and treatment regimen.
Results
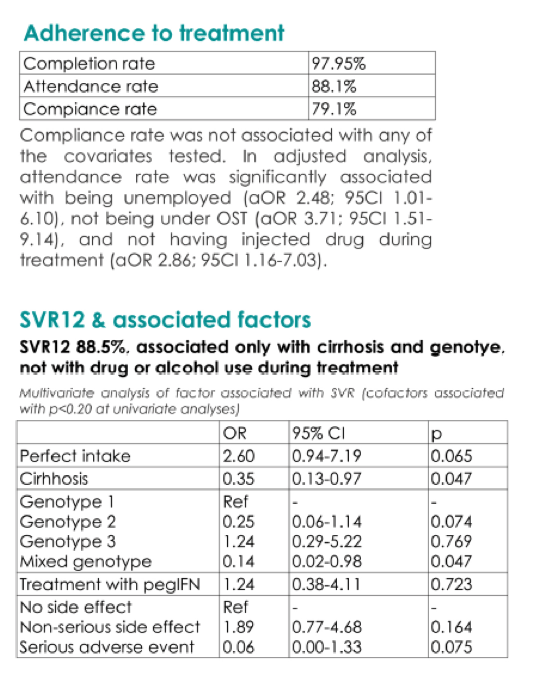
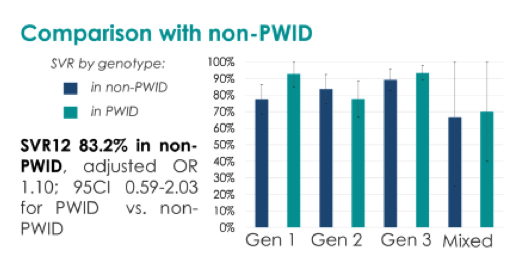
We included 244 PWID in the 2nd semester of 2015. Mean age was 46.3 years, 0.8% were women, 54.5% had cirrhosis (liver stiffness ≥14 kPa or Fib 4>3.25). Genotypes were distributed as follows: 18.9%, 25.4%, 51.6%, and 4.1% for genotype 1, 2, 3 and mixed genotypes, respectively. 2% (n = 5) had to prematurely stop the treatment, due to serious adverse events. Amongst the others, 88.7% never missed any of the bimonthly medical appointments, 79.1% never missed a dose of medication, and 88.2% reached SVR12. Only cirrhosis (adjusted odd ratio (aOR) 0.28; 95% confidence interval (95CI) 0.10-0.83) was significantly associated with SVR12. Ongoing drug use during treatment was associated with delaying medical appointments, but not with observance nor SVR12. SVR12 rate (80.7%) was not significantly different in the 223 non-PWID (aOR 0.94; 95CI 0.50-1.75 compared to PWID) treated at the same time.
Conclusions
In this real life experience, PWID were adherent to care and had SVR12 rates similar to those observed in non-PWID. Concerns about PWID ability to engage in care should not be a reason of exclusion from HCV treatment in Georgia.



| |
| |
| |
|
|
|