 |
 |
 |
| |
MODIFIED ECHO MODEL TO INCREASE ACCESS TO HEPATITIS C TREATMENT:
A HYBRID OF ECHO AND CO-MANAGEMENT
BETWEEN SPECIALTY PROVIDERS AND COMMUNITY PROVIDERS & Rapid Same Day Models of Treatment
|
| |
| |

This model contrasts with one day treatment in USA & in Europe & Australia. Outside UA finger-stick point of-care testing with 60 minutes HCV RNA results is available. Recently FDA modified submission process for testing that appear intended to facilitate approval of these tests for he USA. The only way to approach HCV elimination is approving & funding rapid same-day HCV test & treat & congressional full funding for HCV elimination beyond the current completely inadequate $39 mil a year to CDC (for all hepatitis's) to $400 mill a year for 6 years.
Point-of-care HCV RNA testing with peer-led and nurse-based support to enhance HCV treatment among people with recent injecting drug use at a community-led needle and syringe program: the TEMPO pilot study - see study results below - (11/22/21)
FDA In Brief: FDA Issues Final Orders Reclassifying Certain Hepatitis C Diagnostic Tests from Class III to Class II - improving submission process for HCV point-of-care HCV RNA testing - (11/22/21)
AASLD: HCV-RNA VIRAL LOAD FINGERSTICK ASSAY AS A SIMPLIFIED STRATEGY FOR SCREENING AND LINKAGE TO CARE OF PEOPLE WHO USE DRUGS ATTENDING ITALIAN ADDICTION TREATMENT CENTRES: A PILOT PROJECT - (11/15/21)
AASLD: Rapid Hepatitis C Treatment Initiation in Young People Who Inject Drugs: Final Results from the HCV-Seek, Test & Rapid Treatment (HCV-ST&RT) Randomized Pilot Clinical Trial - (11/15/21) in NYC
AASLD: USE OF SOFOSBUVIR/VELPATASVIR IN TEST AND TREAT STRATEGY IN FRANCE: SNAPSHOT ANALYSIS OF PROSPECTIVE COHORT STUDY HELIOS 3 - (11/12/21)
AASLD: A COMMUNITY-BASED TRIAL OF HEPATITIS C TREATMENT AT POINT OF DIAGNOSIS FOR MARGINALIZED POPULATIONS: PRELIMINARY RESULTS FROM THE NO ONE WAITS (NOW) STUDY - (11/16/21)
AASLD: HCV CASCADE OF CARE [Big Gaps] AND NEXT STEPS FOR HCV ELIMINATION IN THE UNITED STATES FOLLOWING THE COVID-19 PANDEMIC - new updated improved HCV screening guidelines needed including for pregnant women & high-risk populations (11/22/21)
AASLD: HEPATITIS C TREATMENT IN THE UNITED STATES, 2014-2020 numbers of treated is "below that needed to ensure elimination in USA by 2030, increased screening needed, remove all rstrictions" - (11/17/21)

AASLD 2021 Nov 12-15
Nazanin Hooman1, Courtney Hammill1, Alyssa Miller1, Marressa Starks-Baker1, Elaine Milem1, Megan Cooper2, Stromberg Arnold3, Jeffrey Weiss4 and Jens Rosenau1, (1) GI, University of Kentucky, (2)University of Kentucky, (3) Statistics, University of Kentucky, (4)Division of General Internal Medicine, Icahn School of Medicine at Mount Sinai
Background: The ECHO (Extension for Community Health Outcomes) Model has been successfully used to educate community providers through case-based learning on local hepatitis C (HCV) management. Co-management models have been used to help community providers manage HCV with support of a specialty team. We investigated the effectiveness of a novel hybrid model combining the ECHO approach with HCV management support tools (Fibroscan, dedicated specialty pharmacists, social worker/patient navigator team).
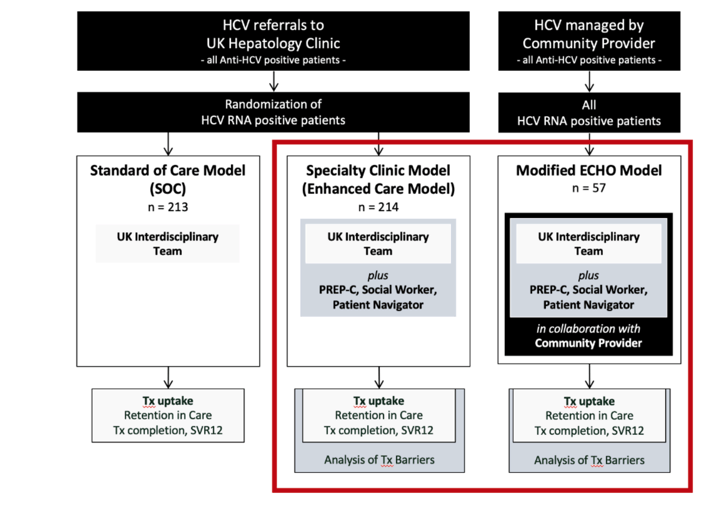
Methods: Patients enrolled in the hybrid ECHO model were referred for a single HCV assessment to the University of Kentucky Hepatology clinic (visit with specialty provider, pharmacist, social worker/patient navigator contact with PREP-C assessment, Fibroscan). Assessment reports were sent to referring providers, and cases were discussed in weekly ECHO sessions. Treatment was executed by community providers with support of the HCV care team. Outcomes were compared with those of patients entirely managed in the clinic (specialty model with equivalent support tools) during the same period. Primary endpoint was HCV treatment uptake within one year after the specialty visit; secondary endpoints were treatment completion and SVR. Analysis was performed using Chi-square, uni- and multivariable logistic regression, and Kaplan-Meier time to event analysis.
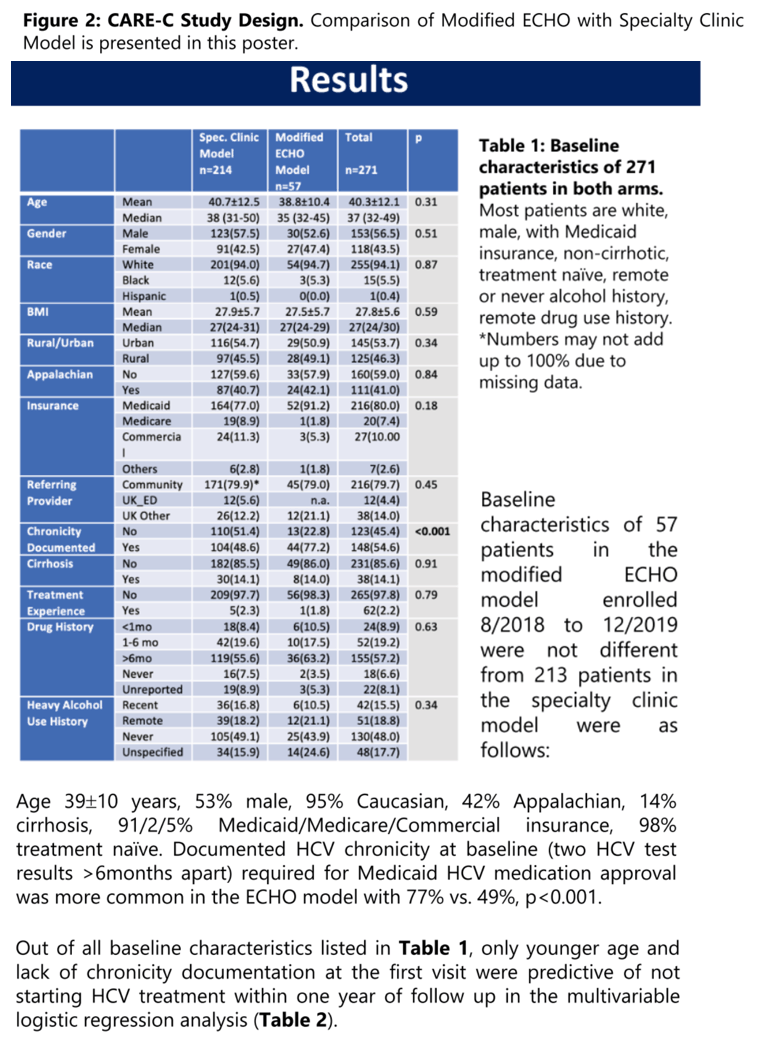
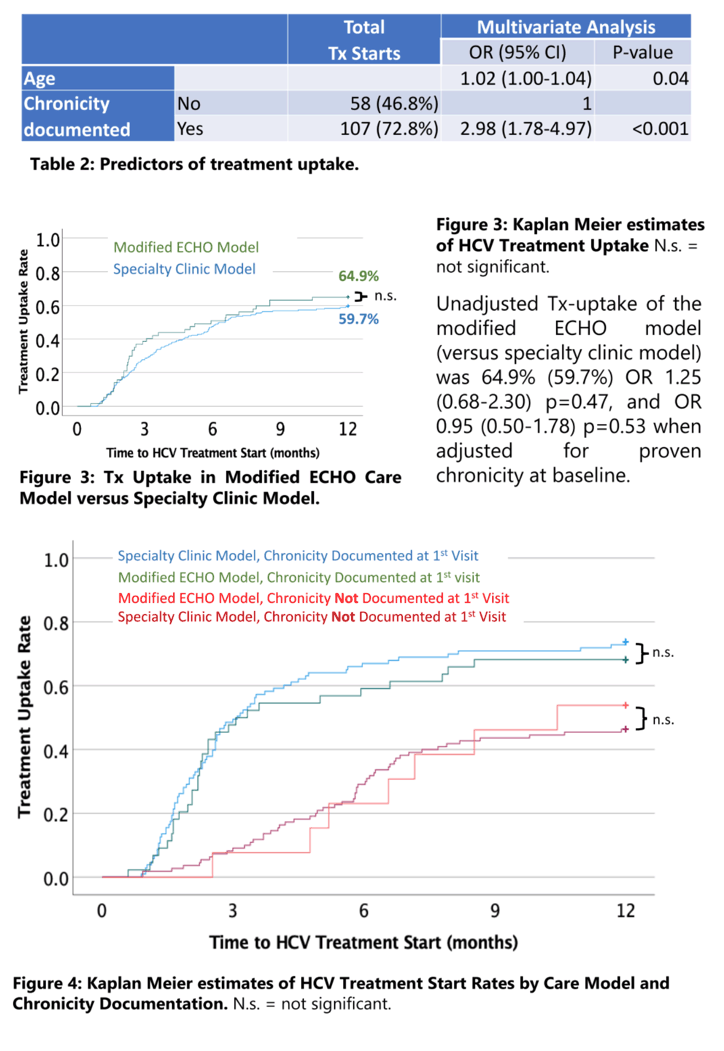
Results: Baseline characteristics of 57 patients in the modified ECHO model enrolled 8/2018 to 12/2019 were not different from 213 patients in the specialty clinic model: Age 39±10 years, 53% male, 95% Caucasian, 41% Appalachian, 14% cirrhosis, 91/2/5% Medicaid/Medicare/Commercial insurance, 98% treatment naïve. Documented HCV chronicity at baseline (two HCV test results >6months apart) required for Medicaid medication approval was more common in the ECHO model with 77% vs. 49%, p<0.001. Unadjusted Tx-uptake of the modified ECHO model (versus specialty clinic model) was 64.9% (59.6%) OR 1.25 (0.68-2.30) p=0.47, and OR 0.95 (0.50-1.78) p=0.53 when adjusted for proven chronicity at baseline. Treatment completion and SVR 12 rates were 70.3% (78.7%) OR 0.64 (0.28-1.45) p=0.29, and 67.6% (76.4%) OR 0.65 (0.29-1.45) p=0.30.
Conclusion: The novel hybrid ECHO model was similar to the specialty clinic model in terms of treatment uptake, completion, and SVR. These findings demonstrate the feasibility and effectiveness of a single visit in the tertiary care center and subsequent collaborative HCV treatment by community providers.


|
| |
|
 |
 |
|
|