 |
 |
 |
| |
HCV Prior Authorization & Elimination Barriers - One Small Step for Rhode Island Medicaid, One Giant Leap Towards Hepatitis C Elimination
|
| |
| |
"DAA makers provide substantial discounts to Medicaid and pharmacy benefits managers. While DAA wholesale acquisition costs have dropped dramatically since sofosbuvir approval, lack of transparency about current pric-ing and discounts continues. I estimate RI's current Medicaid price for eight weeks of glecaprevir/pibrentasvir at $10,000.
On Aug. 1, 2021, Rhode Island (RI) Medicaid removed prior authorizations (PAs) for two direct-acting antiviral (DAA) regimens for hepatitis C virus (HCV), glecaprevir/pibrentasvir and sofosbuvir/velpatasvir/voxilaprevir, following seven other states. Removing PAs will increase access to HCV cure for thousands of RI Medicaid beneficiaries.
This step represents the culmination of eight years of advocacy to ensure equitable access to these life-saving medications that can safely cure 99% of HCV infections in eight to 12 weeks. During these years, for many untreated Rhode Islanders, hepatic fibrosis progressed. Some died unneces-sarily of HCV. Transmission skyrocketed, creating greater medical and economic burdens.11
The arduous PA remained a barrier. This pre-approval process to determine if a patient met payer-specific DAA criteria differed across four RI Medicaid plans (and seven other payers), each with unique requirements, taking 45-120 minutes per patient.19 We completed and faxed a payer-specific PA document, plus laboratory results, to each plan. PAs required repeat blood tests - HCV RNA and genotype within 90 days even for patients with documented viremia for years and recent genotyping, prescribed pan-ge-notypic regimens - and myriad administrative elements (phone calls, peer-to-peer discourse, denials, appeals). Each plan dictated a preferred pharmacy, some mail order only, and a preferred formulary. Many practices lacked staff for this. The process delayed treatment initiation, prevented test-to-treat strategies, and contributed to loss to follow-up.
And oh, the administrative waste. How much time did we spend on this bureaucratic task rather than with patients? The U.S. spends more on health care than any other nation, with the cost of waste accounting for 25%-30% of total health care spending.20-21 The administrative complexity category is associated with the greatest contribution to waste, even more than the inflation of medication pricing.20
There was collateral damage. PAs contributed to misperceptions about HCV. Some clinicians interpreted fibrosis documentation to mean that they could not treat without radiologic evaluation of liver stiffness (not the case). Others inferred that they could not treat people who use drugs, while evidence demonstrates comparable cure rates with and without substance use with appropriate supports, and that reduction in the viremic pool is imperative. Appreciation of the opportunity for quantum progress was lost."
In part due to this history, RI's HCV prevalence exceeds national averages.23 RI joins seven states projected to eliminate HCV latest, beyond 2050.8 It will be said that Medicaid programs, caring for a disproportionate share of HCV, struggle to treat their enrollees. While rationing and an onerous PA pathway curbed DAA prescriptions, RI Medicaid's largest provider recorded $1,366,000,000 revenue in 2017, 35.1% growth percentage, and $354,625,064 revenue growth, as RI's second fastest growing private company.24 It is well established that universal, immediate DAA treatment in Medicaid populations is cost effective and decreases downstream costs, due to avoidance of liver-related complications (not including prevention benefits which decrease risks to the community). As untreated persons reach age 65, costs are shifted to Medicare. Here, medication discounts do not exist, but universal treatment does nationally, increasing DAA expenses for the population not treated earlier.10
---------------------------------------
The key NOW to elimination is approval for rapid 60-minute finger-stick point-of-care test, which is used in Europe & Australia, here are some studies from AASLD last week on these. And of course the federal government & Congress have not been convinced to provide more than $39 mill to the CDC for hepatitis. We need $400 mill year for 6 years to eliminate HCV, the CDC says. PLUS at AASLD all the trend are going in the wring direction: increased HCC rates, treatment rates are down & not adequate to get to elimination, and screening rates are no where near that needed - all because we do not have a fully funded federal HCV Elimination Program
Point-of-care HCV RNA testing with peer-led and nurse-based support to enhance HCV treatment among people with recent injecting drug use at a community-led needle and syringe program: the TEMPO pilot study - see study results below - (11/22/21)
FDA In Brief: FDA Issues Final Orders Reclassifying Certain Hepatitis C Diagnostic Tests from Class III to Class II - improving submission process for HCV point-of-care HCV RNA testing - (11/22/21)
AASLD: HCV-RNA VIRAL LOAD FINGERSTICK ASSAY AS A SIMPLIFIED STRATEGY FOR SCREENING AND LINKAGE TO CARE OF PEOPLE WHO USE DRUGS ATTENDING ITALIAN ADDICTION TREATMENT CENTRES: A PILOT PROJECT - (11/15/21)
AASLD: Rapid Hepatitis C Treatment Initiation in Young People Who Inject Drugs: Final Results from the HCV-Seek, Test & Rapid Treatment (HCV-ST&RT) Randomized Pilot Clinical Trial - (11/15/21) in NYC
AASLD: USE OF SOFOSBUVIR/VELPATASVIR IN TEST AND TREAT STRATEGY IN FRANCE: SNAPSHOT ANALYSIS OF PROSPECTIVE COHORT STUDY HELIOS 3 - (11/12/21)
AASLD: HCV CASCADE OF CARE [Big Gaps] AND NEXT STEPS FOR HCV ELIMINATION IN THE UNITED STATES FOLLOWING THE COVID-19 PANDEMIC - new updated improved HCV screening guidelines needed including for pregnant women & high-risk populations (11/22/21)
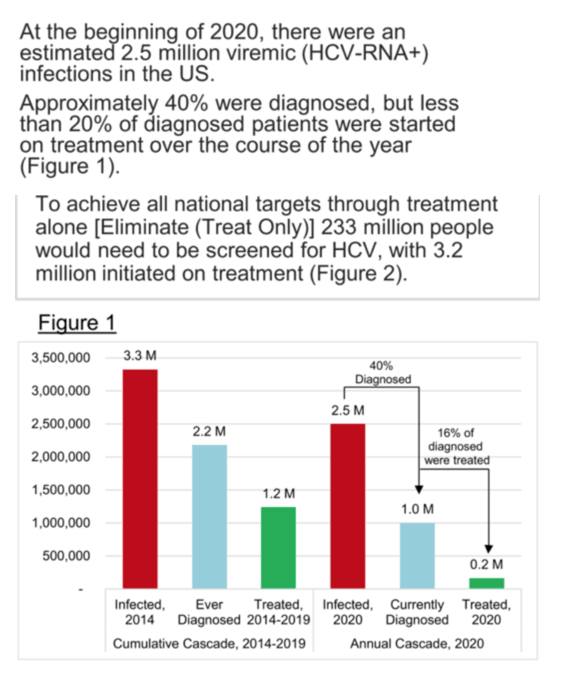
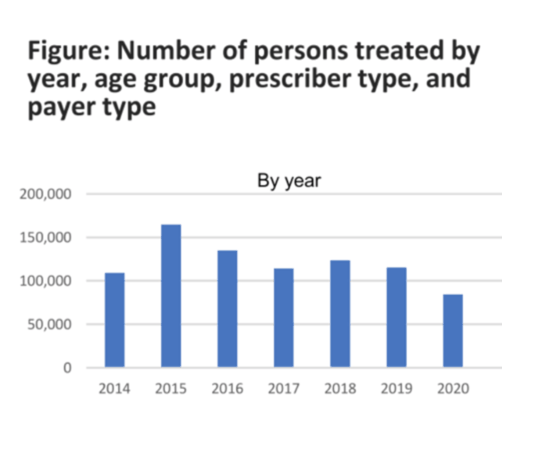
AASLD: HEPATITIS C TREATMENT IN THE UNITED STATES, 2014-2020 numbers of treated is "below that needed to ensure elimination in USA by 2030, increased screening needed, remove all rstrictions" - (11/17/21)
During 2014-2020, an average of 120,000 persons with hepatitis C were treated with DAAs annually. This average number of persons living with hepatitis C treated is below the number needed to ensure elimination of hepatitis C as a public health threat in the United States by 2030. Public health interventions such as increased testing and improved linkage to care for groups experiencing disproportionate impact, and removal of restrictions
AASLD: POINT-OF-CARE TESTING FOR HEPATITIS C IN PUBLIC CLINICAL LABORATORIES REVEALS SIGNIFICANT RATES OF HIDDEN HCV AMONG ELDERLY POPULATION IN SOUTHERN ITALY (12/01/21)
|
| |
|
 |
 |
|
|