 |
 |
 |
| |
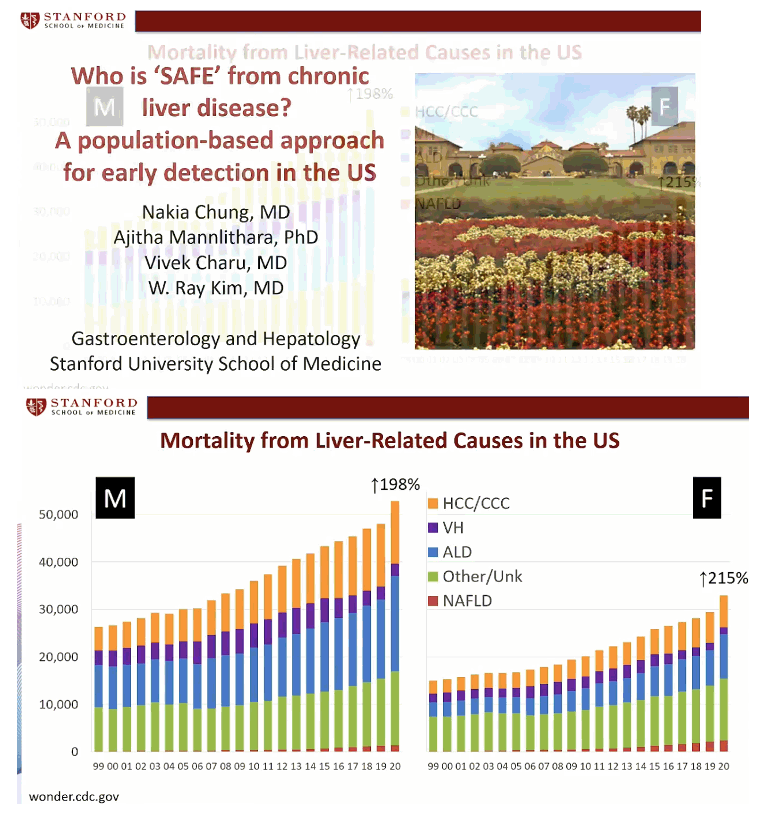
SAFE Score Rates Liver Disease Risk Well in US General Population - Who is SAFE from chronic liver disease? A population-based approach for early detection in the US.
|
| |
| |
EASL Congress 2023, June 21-24, Vienna
Download the PDF here
Mark Mascolini
Originally devised to gauge risk of liver disease progression in people with nonalcoholic fatty liver disease (NAFLD), the SAFE score worked well in stratifying people in the US general population into low, intermediate, or high liver disease risk clusters [1]. Researchers from Stanford University who conducted this study hope their findings will help primary care clinicians accurately gauge liver disease risk so they can continue caring for people at lower risk whom they might now refer because of incidental findings like above-normal alanine aminotransferase (ALT). The Stanford team published some of these findings earlier this year [2].
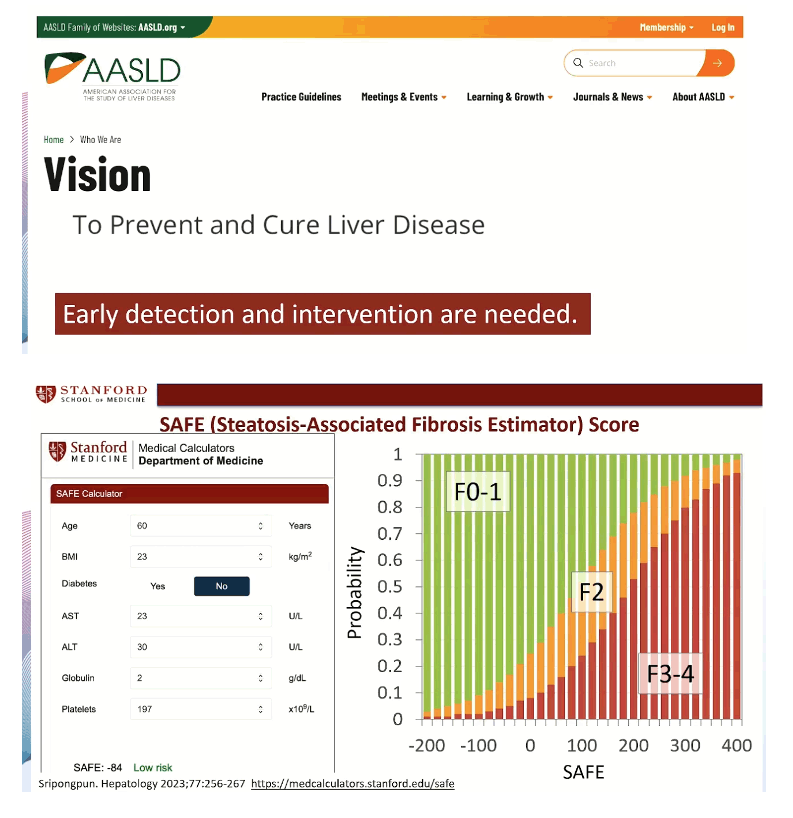
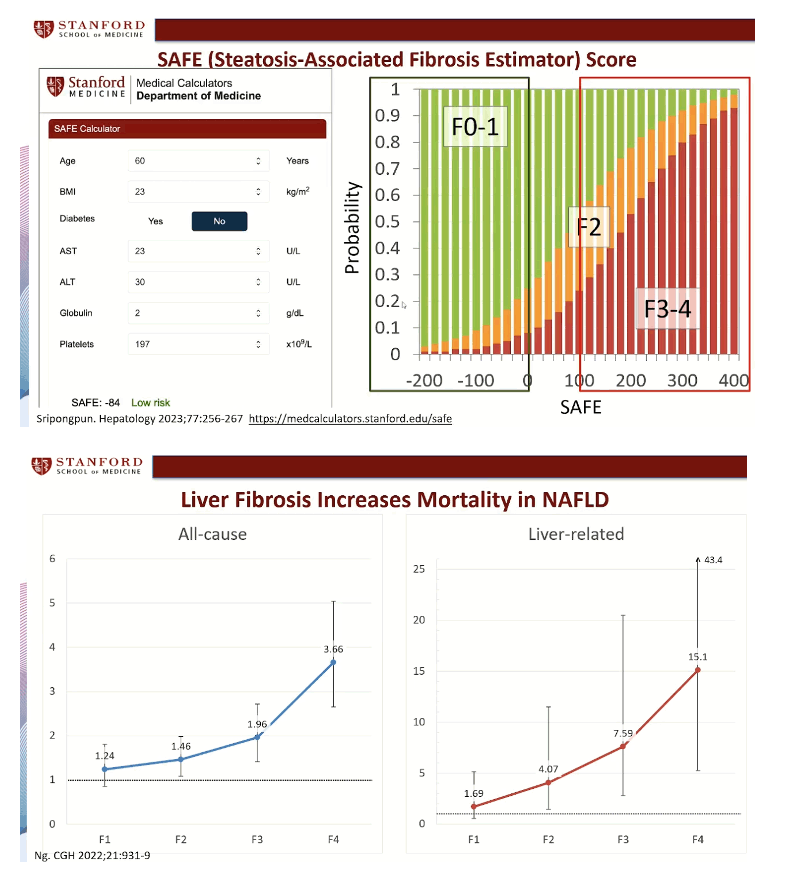
SAFE, which stands for steatosis-associated fibrosis estimator, includes 7 easily obtained variables: age, body mass index, diabetes status, aspartate aminotransferase (AST), ALT, globulin (total serum protein minus albumin), and platelets. An online SAFE calculator appears on the Stanford website: https://medcalculators.stanford.edu/safe. If the calculator yields a score below 0, that person has a low risk of clinically significant fibrosis, equivalent to fibrosis stages F0 and F1. A score between 0 and 99 means an intermediate risk, equivalent to F2 fibrosis, and a score of 100 or higher signals a high risk of clinically significant fibrosis, equivalent to F3 or F4. Meta-analysis by other researchers showed that F2 fibrosis stands at an inflection point in the ascending mortality curves for liver-related and all-cause mortality [3].
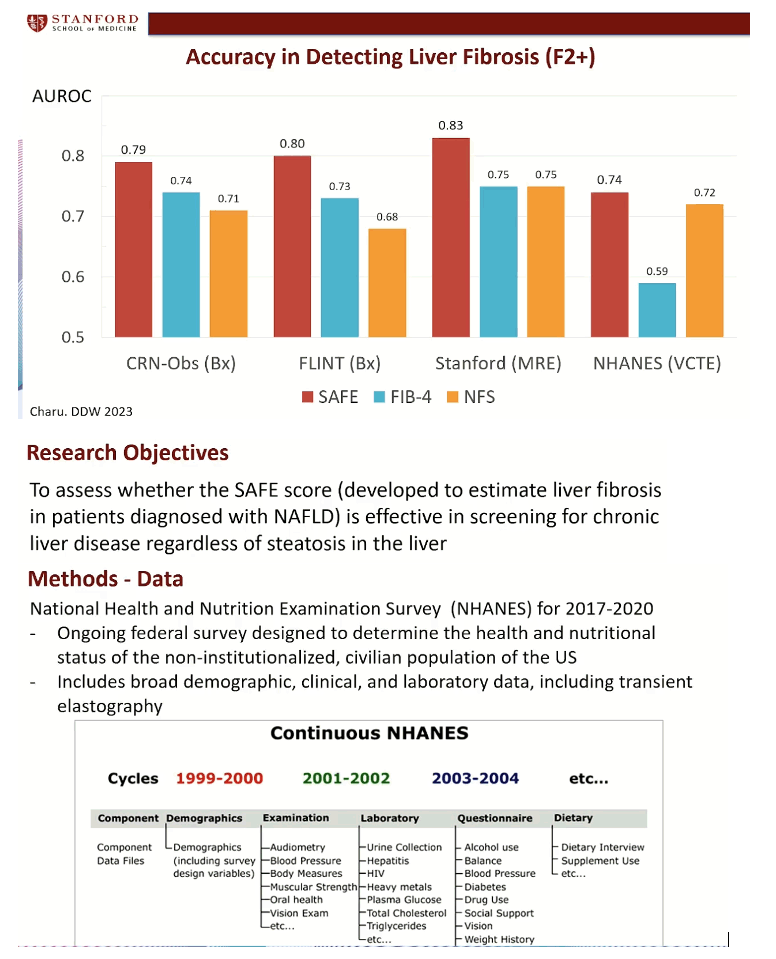
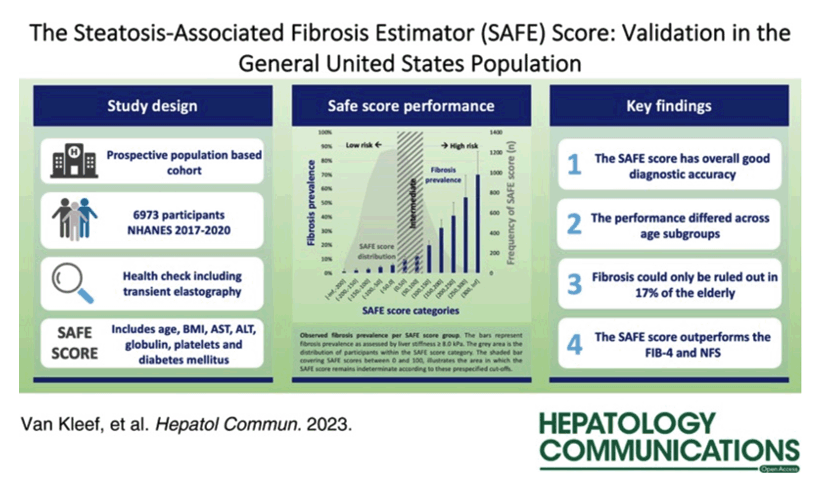
Using data from 676 people in the nonalcoholic steatohepatitis (NASH) Clinical Research Network observational study, the Stanford investigators applied logistic regression and machine learning to build prediction models that separated F2 or worse fibrosis from F0 or F1 fibrosis. They calculated an area under the receiver operating characteristic (AUROC) curve showing that the selected model (SAFE) predicted F2 or worse fibrosis with 79% accuracy, compared with 74% when they used FIB-4 to predict fibrosis stage and 71% when they used the NAFLD fibrosis score (NFS).
Next the researchers validated this model in 280 people in the FLINT trial and 130 local people with NAFLD who had magnetic resonance elastography data. Accuracy in detecting F2 or worse fibrosis proved consistently greater with SAFE than with FIB-4 or NFS in the FLINT trial population (AUROC 80%, 73%, 68%) or the Stanford elastography group (AUROC 83%, 75%, 75%). The final model proved a better predictor of F2 or worse fibrosis in 11,953 people in a large US observational study, the National Health and Nutrition Examination Survey (NHANES) III (AUROC 74%, 59%, 72%).
The new part of the Stanford study aimed to see whether SAFE, which was developed to estimate liver fibrosis in people with diagnosed NAFLD, also works in screening for chronic liver disease in the US population at large, regardless of liver steatosis status. They once more delved into NHANES data for the period 2017-2020, which includes noninstitutionalized civilian US residents and gathers broad demographic, clinical, and lab data, including transient elastography. The researchers stratified people into SAFE tiers indicating low risk (score below 0), intermediate risk (0 to 99), or high risk (100 or higher) of clinically significant fibrosis.
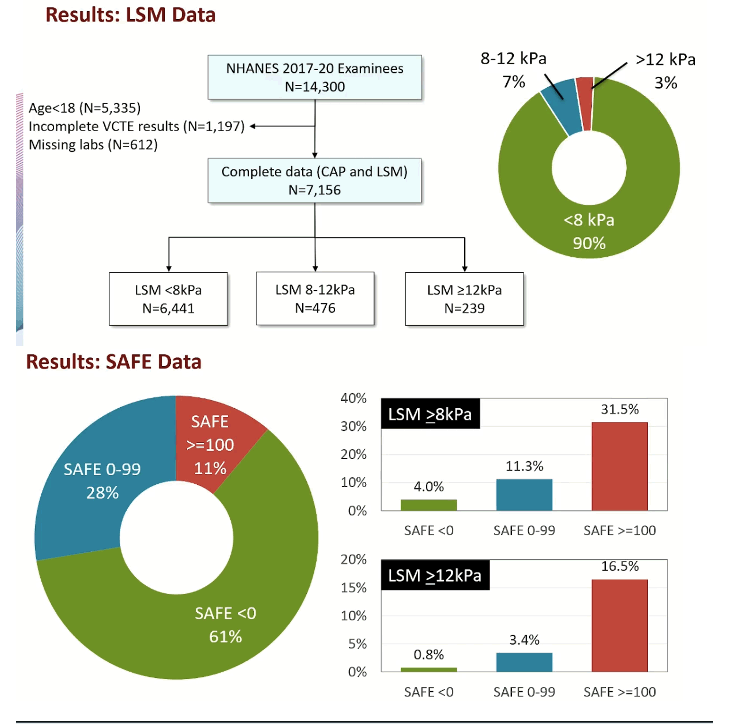
After eliminating NHANES participants younger than 18 or missing lab data, the investigators wound up with 7156 people, 6441 (90%) with a transient elastography liver stiffness measure (LSM) below 8 kPa (consistent with F0-F1 fibrosis), 476 (7%) with LSM at 8 to 12 kPa (equivalent to F2 fibrosis), and 3% with LSM above 12 kPa (consistent with F3 or worse fibrosis).
SAFE scores in these NHANES participants stood below 0 in 61%, at 0 to 99 in 28%, and at 100 or more in 11%. A higher SAFE score boosted the probability of clinically significant fibrosis: For people with LSM at or above 8 kPa, only 4% had a SAFE score below 0, while 11.3% had a 0-99 SAFE score and 31.5% had a 100-or-greater SAFE score. For people with LSM at or above 12 kPa, only 0.8% had a SAFE score below 0, while 3.4% had a SAFE score of 0-99 and 16.5% had a SAFE score of 100 or more.
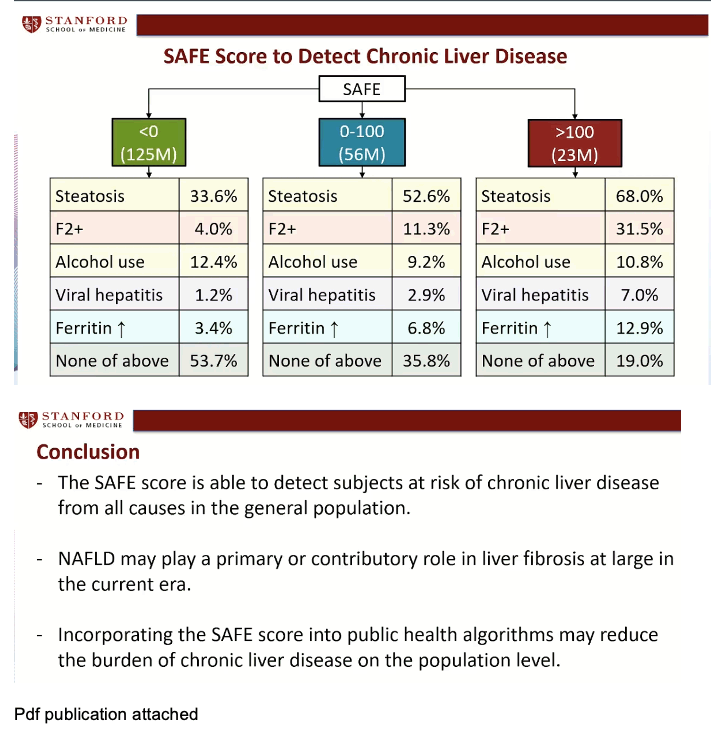
Applying these findings to the US general population, the Stanford group projected that steatosis would account for liver disease in 33.6% of 125 million people with a SAFE score below 0. In contrast, steatosis would affect 52.6% of 56 million people with a SAFE score of 0 to 99, and 68.0% of 23 million with a SAFE score of 100 or more. Respective rates of F2 or worse fibrosis were 4.0%, 11.3%, and 31.5%, and of viral hepatitis 1.2%, 2.9%, and 7.0%.
The Stanford team argued that primary care use of the SAFE scoring method (https://medcalculators.stanford.edu/safe) could shift liver disease management in the US from a specialty-driven approach to a population-based approach. In the specialty-driven paradigm, a primary care clinician might notice a repeated high ALT and refer that person to a hepatologist. But the primary care clinician could manage many such individuals without specialist input. The SAFE tool, the researchers proposed, would allow clinicians to stage and stratify more people with liver disease signals and refer only those who truly need specialist care.
References
1. Chung N, Mannalithara A, Charu V, et al. Who is SAFE from chronic liver disease? A population-based approach for early detection in the US. EASL Congress 2023, June 21-24, Vienna. Abstract OS-115-YI.
2. Sripongpun P, Kim WR, Mannalithara A, et al. The steatosis-associated fibrosis estimator (SAFE) score: A tool to detect low-risk NAFLD in primary care. Hepatology. 2023;77:256-267. doi: 10.1002/hep.32545. https://journals.lww.com/hep/Abstract/2023/01010/
The_steatosis_associated_fibrosis_estimator__SAFE_.24.aspx
3. Ng CH , Hui Lim WH, Lim GEH, et al. Mortality outcomes by fibrosis stage in nonalcoholic fatty liver disease: a systematic review and meta-analysis. Clin Gastroenterol Hepatol. 2023;21:931-939.e5. doi: 10.1016/j.cgh.2022.04.014. https://www.cghjournal.org/article/S1542-3565(22)00439-6/fulltext

in the elderly, the specificity was poor (18% low cutoff and 71% high cutoff).
Moreover, of the total clinically significant fibrosis cases (n=134) in the young group, 89 (66%) resided in the SAFE<0 (ruled out) group and were thus missed.
In general, the FIB-4 has low predictive value, even among the middle aged (40-60 y) for which it was designed, whereas the NFS had better performance across subgroups. However, the NFS had (similar to the SAFE score) great differences in test characteristics across age groups while maintaining reasonable AUCs. Moreover, the NFS, like the SAFE score, had a large proportion (30%) in which the fibrosis risk remained indeterminate using the prespecified cutoffs of <-1.455 for ruling out and >0.675 for ruling in fibrosis. Finally, similar to the SAFE score, the high-risk group, according to the FIB-4 and NFS, increased substantially with age, disconcordant with the clinically significant fibrosis prevalence.
We investigated the performance of the SAFE score in the detection of patients at high risk of clinically significant fibrosis in the general population and demonstrated that the SAFE scorehas an overall good performance, especially among middle-aged individuals (aged 40-60) but lacks performance in younger (aged 18-40) and the ability to rule out clinically significant fibrosis in older (aged 60-80) populations. The test characteristics were comparable in target populations with metabolic dysfunction or steatosis, in which noninvasive assessment of fibrosis is indicated.
|
| |
|
 |
 |
|
|