 |
 |
 |
| |
A Phase 2a, randomized, active-comparator-controlled, open-label study
to evaluate the efficacy and safety of efinopegdutide in individuals with
nonalcoholic fatty liver disease
|
| |
| |
EASL 2023 June 23 Vienna
Manuel Romero-Gómez1, Eric Lawitz2, R. Ravi Shankar3, Eirum Chaudhri3, Jie Liu3, Raymond Lam3, Keith D. Kaufman3, Samuel Engel3
1University of Seville/ Virgen del Rocio University Hospital, Institute of Biomedicine of Seville (HUVR/CSIC/US)/ Digestive Diseases Unit and CIBERehd, Sevilla, Spain; 2Texas Liver Institute, University of Texas Health San Antonio, San Antonio, TX, USA; 3MRL, Merck & Co., Inc., Rahway, NJ, USA

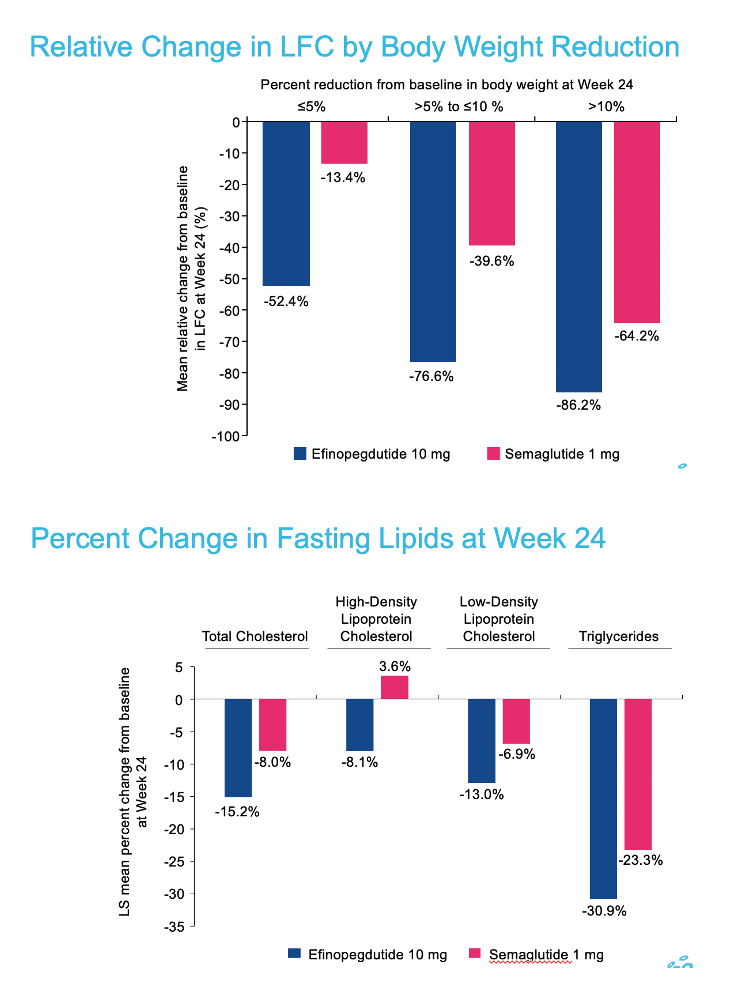
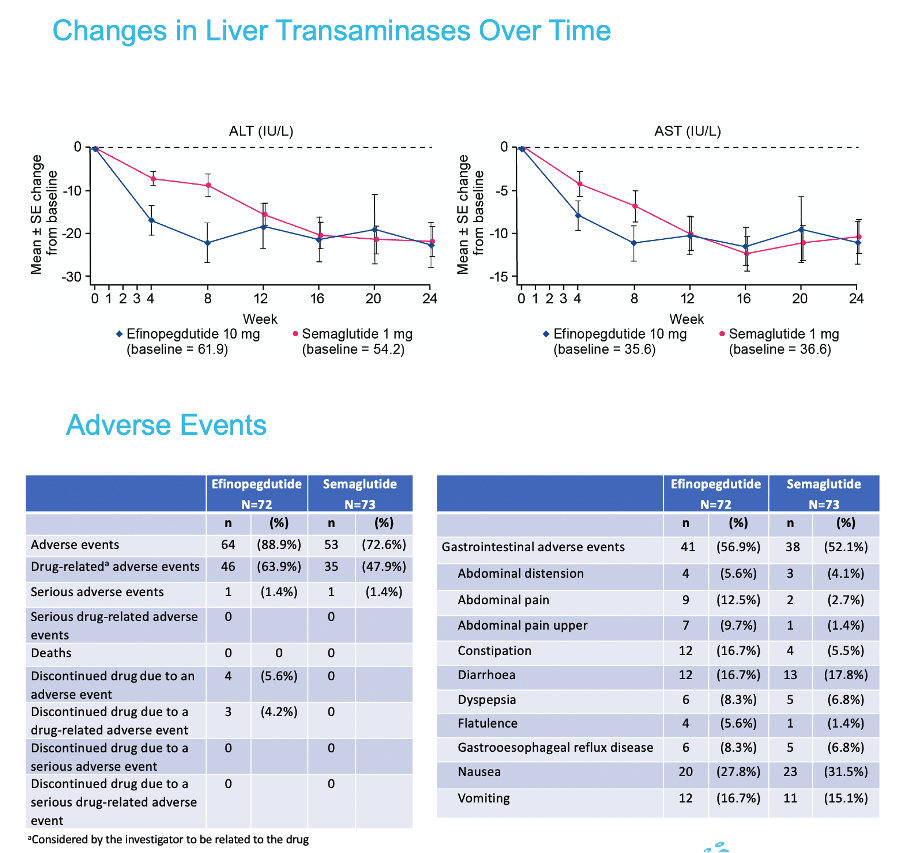
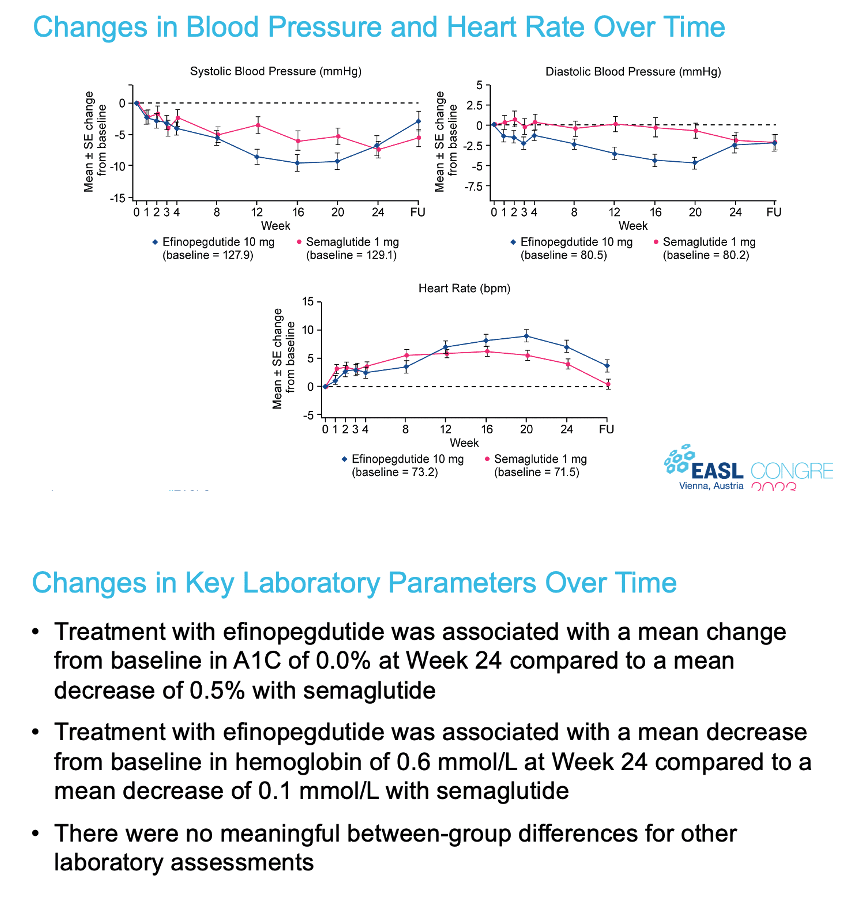

|
| |
|
 |
 |
|
|