| |
Neurocognitive Dysfunction; Premature Cognitive Aging in PLWH Adjusting for Comorbidities & Comorbidities Contribute
|
| |
| |
Cognitive impairment in aging older PLWH in this study from Australia is identified as prematurely onsetting in well educated gay men & of note they adjusted for comorbidities suggesting its worse for others with more bad predictors including those with more comorbidities, African-Americans, HIV+ women, Latinos, drug abuse history, mental health disorder history. I think its clear the problem of cognitive impairment is understated & underappreciated & I think will emerge as a much worse problem as large numbers of PLWH age into their 70s.
Jules Levin
NATAP
"Our study supports that PLHIV aged ≥50 years should undergo regular neurocognitive and mental health screen and further investigation when this screenis positive [66]. However, implementation research is urgently needed to enable a realistic clinical translation."
"A potential explanation for premature cognitive ageingin HIV includes chronic immune activation and immunesenescence [53,54]. This mechanism is similar to what has been found in the normal ageing process, so that it has been postulated that the ageing phenomenon start searly among PLHIV [55]. Importantly, there is evidence of chronic neuroimmune activation and associated neuroinflammation despite successful cART and viral sup-pression [56]. Altogether, ageing and HIV may lead to brain damage via excitotoxicity, mitochondrial dysfunction and oxidative stress [53,57]."
CROI: NEUROCOGNITIVE DYSFUNCTION, INFLAMMATION, AND ADIPOSITY IN TREATED HIV PATIENTS - (03/24/21)
Motor function declines over time in human immunodeficiency virus and is associated with cerebrovascular disease, while HIV-associated neurocognitive disorder remains stable - (02/26/21)
yes, comorbidities contribute to cognitive impairment with aging for PLWH
CROI: HIGHER COMORBIDITY BURDEN PREDICTS WORSENING NEUROCOGNITION IN PEOPLE WITH HIV - (03/11/21)
-------------------------------------
4 Studies on Cognitive Aging in PLWH....
- the 1st study immediately below from Australia of gay well educated men found premature cognitive aging; men >50 had worse predictors...& of note the authors reported adjusting for comorbidities......
the 2nd study below reports decline in motor function over time in PLWH.....the 3rd study from CROI 2021 found worse cognitive function for HIV+ including worse reaction time and speed processing and it was associated with inflammation markers and higher body fat composition.
Cognitive ageing is premature among a community sample of optimally treated people living with HIV - (02/26/21)
Neurocognitive function was evaluated with the Cogstate Computerized Battery (CCB) at baseline and 6 months after. Linear mixed effects (LME) models examined main and interaction effects of HIV status and chronological age on the CCB demographically uncorrected global neurocognitive score (GZS), adjusting for repeated testing, and then adjusting sequentially for HIV disease markers, mental health and comorbidities.
The main study finding was evidence of premature cognitive ageing. The current study identified premature cognitive ageing in a community sample of well‐educated and well‐treated HIV‐positive gay and bisexual men with low AIDS rate, compared with demographically comparable HIV‐negative counterparts. The final model that was restricted to the HIV-positive participants is presented in Table 5. Older age (b=0.32, 95% CI:0.45 to 0.19, P<0.001) was associated with lower GZS. Having a history of non-HIV neurological condition (b=0.11, 95% CI:0.22 to 0.001, P<0.05), having been diagnosed with CDC stage C (b=0.10, 95% CI:0.21 to0.02,P<0.05), and having a higher anxiety score (b=0.17, 95% CI:0.25to0.08,P<0.001) were also associated with a lower GZS. In addition, compared with baseline testing, cognitive testing at 6 months was negatively associated withlower GZS among HIV-positive participants (b=0.05,95% CI:0.11 to0.03,P<0.05). On the other hand, duration of ART (b=0.19, 95% CI: 0.004–0.37,P<0.05) was associated with a higher GZS. CDC stage C implies that PLHIV may have experienced high viral load and/or low nadir CD4 and/or an AIDS-defining illness. As our analysis also included nadir and current CD4 counts, as well as viral load, it suggests that the CDC stage may be the strongest predictor for NCI.
The main study finding was evidence of premature cognitive ageing. Essentially, the HIV and age interaction effect size was the largest (medium effect size) compared with all the variables that were significantly associated with neurocognitive performance. Several factors were also independently associated with neurocognitive performance, although they all had small effect sizes. In the analyses including both HIV-negative and HIV-positive samples, a higher degree of anxiety symptoms, a history of HIV-related brain involvement, and a past CDC stage C diagnosis were associated with lower neurocognitive performance, while SUD was associated with higher cognitive performance. In the analyses restricted to the HIV-positive sample, in addition to age, which has a small to medium effect size, a history of non-HIV-related neurological disorder, a past CDC stage C diagnosis and a shorter duration of ART were associated with poorer neurocognitive performance, all representing small effect sizes.
Figure 1 presents the interaction effect of HIV and age on cross-sectional GZS predicted by the regression model.It is evident that neurocognitive performance deteriorated with increasing age at a significantly higher rate amongHIV-positive than among HIV-negative participants.
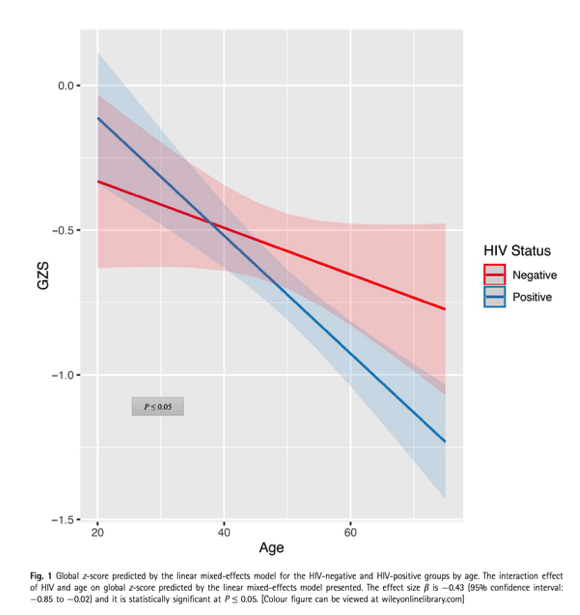
This study enrolled 254 PLHIV [92% on antiretroviral therapy; 84% with HIV RNA<50 copies/mL; 15% with AIDS) and 72 HIV-negative gay and bisexual men [mean (SD) age=49 (10.2) years]from a single primary care clinic in Sydney, Australia. Neurocognitive function was evaluated withthe Cogstate Computerized Battery (CCB) at baseline and 6 months after. Linear mixed-effects(LME) models examined main and interaction effects of HIV status and chronological age on the CCB demographically uncorrected global neurocognitivez-score (GZS), adjusting for repeated testing, and then adjusting sequentially for HIV disease markers, mental health and comorbidities.
Figure 1 presents the interaction effect of HIV and age on cross-sectional GZS predicted by the regression model. It is evident that neurocognitive performance deteriorated with increasing age at a significantly higher rate amongHIV-positive than among HIV-negative participants.
With ageing, neurological and psychological health is becoming increasingly relevant for PLHIV. First, cognitiveageing may be premature among PLHIV because of persistent immune activation and inflammation along withan increased prevalence of other age-related comorbidi-ties [especially cardiovascular diseases (CVD) and stroke],all of which are known risk factors for dementia in thegeneral population [6]. Second, age is the number onerisk factor for cognitive decline and dementia in the gen-eral population [7] and even mild evidence of prematurecognitive ageing could have serious public health impli-cations for the ageing HIV epidemic. Third, PLHIV carrya significant mental health burden such as anxiety anddepression [8,9], which are also known risk factors forcognitive decline and dementia [10,11].
Our review [12] also demonstrated that only a minorityof cognitive ageing studies adjusted for the effects ofage-related comorbidities and other comorbidities thatmay affect neurocognitive performance. Indeed, in addi-tion to the effect of chronological age, an array of otherfactors such as HIV clinical characteristics (e.g. clinicalstage, nadir and current CD4 counts and HIV viral load),previous HIV brain involvement [i.e. historical centralnervous system (CNS) opportunistic infection (OI) andHIV-associated neurocognitive disorder (HAND)], age-re-lated comorbidities, mental health burden and lifestylefactors will complicate the degree to which prematureageing can be ascribed [16,17].
To assess our primary aim, the study participants were divided a prior iinto four groups by age category andHIV status (younger,<50 years; older, ≥50). This cut-off of 50 years old was used based on previous evidence that this age represents a clinically meaningful cut-off in HIV and ageing research:
- Older HIV-positive participants had lower nadir and current CD4 T cell counts, higher ART uptake, and longer HIV duration compared with the youngerHIV-positive participants atP<0.05.
- The older HIV-positive participants had the lowest performance on the GZS at both baseline and follow-up compared with all other groups.
- HAND/NCI prevalence was significantly higher among HIV-positive than among HIV-negative participants at both baseline (42%vs. 22%) and follow-up (43%vs. 23%) (P<0.05).
- Neither HIV status (b=0.27) nor age (b=0.13) main effect was significantly associated with the GZS atP<0.05. However, their interaction was significantly associated with a lower GZS (b=0.43, 95% CI:0.85 to0.02,P<0.05) on GZS. Three covariates were significantly associated with lower GZS across the study period. These included having a history of HIV brain involvement (b=0.12, 95%CI:0.22 to0.01,P<0.05); having been diagnosedwith CDC stage C (b=0.12, 95% CI:0.22,0.03,P<0.05) and having higher anxiety score (b=0.11,95% CI:0.19 to0.04,P<0.01). By contrast, current SUD was associated with higher GZS in an unexpecteddirection (b=0.07, 95% CI: 0.004–0.14)
• Our study supports that PLHIV aged ≥50 years should undergo regular neurocognitive and mental health screen and further investigation when this screenis positive [66]. However, implementation research isurgently needed to enable a realistic clinical translation.
Results
HIV status and age interacted with a lower GZS (b=0.43,P<0.05). Higher level of anxiety symptoms (b=0.11,P<0.01), historical AIDS (b=0.12,P<0.05) and historical HIV brain involvement (b=0.12,P<0.05) were associated with lower GZS.
Conclusions
We found a robust medium-sized premature ageing effect on cognition in a community sample with optimal HIV care. Our study supports routine screening of cognitive and mental health amongPLHIV aged≥50 years.
Conclusion
The current study identified premature cognitive ageing in a community sample of well-educated and well-treated HIV-positive gay and bisexual men with low AIDS rate,compared with demographically comparable HIV-negative counterparts. The study was conducted at a primary care clinic in Sydney, Australia, and included allcomersin the HIV-positive group. However, the effects of psychiatric, HIV and non-HIV neurological and other medicalcomorbidities were adjusted in the analyses. In terms ofclinical implications, our study supports regular neu-rocognitive screening in treated (even if virally sup-pressed) PLHIV aged≥50 years, particularly in thosewith a historical CDC stage C, or a history of HIV-related and non-HIV-related neurological conditions. Concurrent monitoring and intervention on mental health burden should also be considered for PLHIV to prevent additional risk to neurocognitive decline. In terms of research implications, it is critical that further resources and researchfunding be allocated to the follow-up of such cohorts.
A potential explanation for premature cognitive ageingin HIV includes chronic immune activation and immunesenescence [53,54]. This mechanism is similar to what has been found in the normal ageing process, so that it has been postulated that the ageing phenomenon start searly among PLHIV [55]. Importantly, there is evidence of chronic neuroimmune activation and associated neuroinflammation despite successful cART and viral sup-pression [56]. Altogether, ageing and HIV may lead to brain damage via excitotoxicity, mitochondrial dysfunction and oxidative stress [53,57].
Evidence of premature cognitive ageing in a cohort that is optimally treated and is representative of the multiple comorbidities found in a community sample has serious public health ramifications. As age is the number one factor for all-type dementia, it will be very important to follow this kind of cohort into their 70s,the age at which the dementia risk increases exponen-tially in the general population [6]. More worryingly, atthe global level, because PLHIV who have this level of healthcare and cART access, and low AIDS proportion are in the minority [58], it can be expected that in less healthy PLHIV, the premature ageing effect may be even larger, as would be the dementia risk [13].
The implications of premature ageing are not onlyrelated to dementia risk. Poor cognitive health negatively affects physical and social functioning in ageing PLHIV[59,60]. This, in turn, impacts quality of life [61]. Poor cognitive health may also lead to less cART adherence [62], potentially reducing the high compliance rate among olderPLHIV [63]. Although there is no evidence of this in thecurrent cohort, this is a possibility that cannot be ignored in general. Importantly, cognitive decline remains independently associated with mortality in the cART-treatedcohort [64]. A higher mortality rate in people with HANDor other dementia may falsely lead the HIV research com-munity to think that cognitive issues are less representedthan they actually are due to a survivor effect [65].
The clinical implication of our resultsis that provision of regular mental health support, includ-ing for anxiety, remains critical in ageing PLHIV[38,44,45]. Importantly, stress management and cognitiveb ehavioural therapies have been reported to be effective in alleviating anxiety symptoms among PLHIV [46].
Our finding that having CDC stage C was negativelyassociated with neurocognitive function representscumulative evidence that AIDS is a risk factor for NCI[47]. Heatonet al. [48] also showed, when focusing on CDC stage, that NCI rate increased with the advancing CDC stage in both the pre-cART and cART cohorts.CDC stage C implies that PLHIV may have experienced high viral load and/or low nadir CD4 and/or an AIDS-defining illness. As our analysis also included nadir and current CD4 counts, as well as viral load, it suggests that the CDC stage may be the strongest predictor for NCI.
Motor function declines over time in human immunodeficiency virus and is associated with cerebrovascular disease, while HIV-associated neurocognitive disorder remains stable - (02/26/21)
We found that motor dysfunction, cognitive impairment, and cerebrovascular disease were significantly associated with each other at baseline. Cerebrovascular disease independently predicted cognitive impairment in a multivariable model. Longitudinal analysis in a subset of 78 participants with ≥ 4 years of follow-up showed a stable cognition but declining motor function. We conclude that the HDMS is a valid measurement of motor dysfunction in HIV-infected patients and is associated with cognitive impairment and the presence of cerebrovascular disease. Cognitive impairment is mild and stable in CART-treated HIV; however, motor function declines over time, which may be related to the accrual of comorbidities such as cerebrovascular disease.....it is possible that anincreasing burden of cerebrovascular disease in the agingHIV population is one explanation why motor abnormalitiesstill accompany cognitive dysfunction, even when the mostsevere form (i.e., HAD) is in decline......our longitudinal anal-ysis which showed that although cognition remained general-ly stable over the observation period, motor abnormalities worsened. The lack of significant cognitive decline is an en-couraging finding; however, the accrual of motor dysfunctionis worrisome. Motor dysfunction is part of many neurologicdisorders of aging and has real impact on a person's ability toage successfully and maintain independence (Wallace et al.2017 ). The combination of neuropsychological impairment,even if stable and relatively mild, with motor dysfunctionmay contribute to a frail HIV-infected aging population evenin the setting of immune reconstitution and viral suppression.
CROI: NEUROCOGNITIVE DYSFUNCTION, INFLAMMATION, AND ADIPOSITY IN TREATED HIV PATIENTS - (03/24/21)
This is a cross-sectional study involving 65 HIV+ and 33 matched HIV - controls recruited at University Hospitals Cleveland Medical Center
Compared to controls, HIV+ participants had a lower overall cognitive score (76% vs 83%, p=0.01), and performed poorer across different cognitive domains [visuospatial, memory, executive function, naming/language, delayed recall, and abstraction, (all p<0.05)].
In the HIV+ group, lower cognitive testing domain scores were associated with higher inflammatory markers(IL6, TNFR-I, TNFR-II, hsCRP), and with higher body fat compositions (total percent fat and visceral adipose tissue). Overall, 53% were male, 47% were African American, with a mean age of 43 years. Among HIV + individuals, all were on ART by design and 80% had an undetectable HIV-1 RNA level (≤20 copies/ml).
|
|
| |
| |
|
|
|